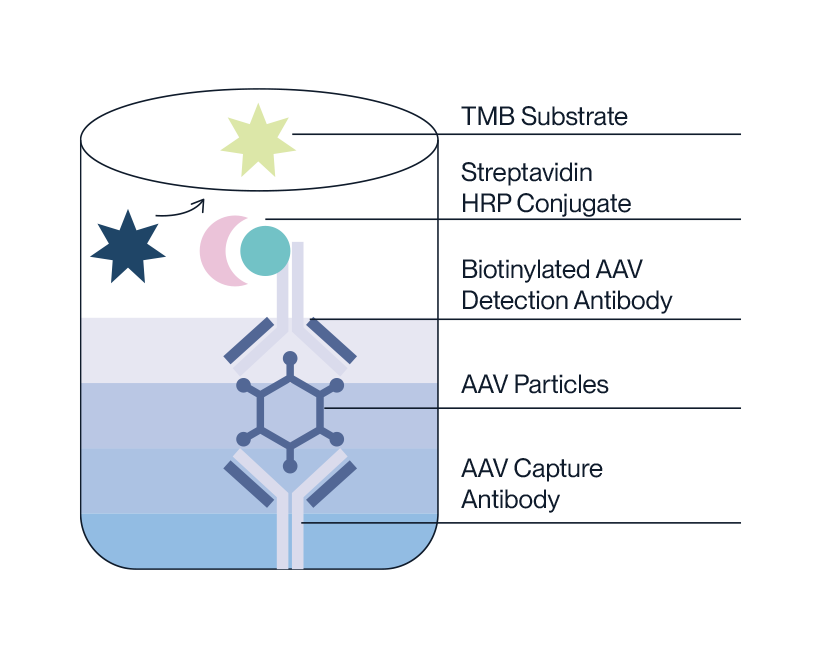
A familiar protocol for easy execution.
Our AAV Titration ELISA kits use a standard sandwich ELISA method utilizing exclusive AAV antibodies developed using our proprietary immunization scheme.
- Pre-coated microtiter plate with removable single-use test strips.
- AAV standard and sample is incubated on plate.
- Biotinylated detection antibody is added to bind AAV capsids.
- Streptavidin HRP conjugate is added to bind with antibody-AAV complex.
- HRP will then oxidize TMB substrate to produce a color change porportional to AAV capsid titer.
Proprietary monoclonal antibodies developed using enhanced immunization scheme.
Our AAV ELISA kits using recombinant monoclonal antibodies developed using our proprietary immunization scheme that overcomes the weak AAV immunogenicity. This method maintains a remarkable level of consistency as the serum titers within the same group of mice consistently align. Across all AAV serotypes, our technique consistently elevates AAV anti-serum titers, resulting in the production of high- performance monoclonal antibodies.
Browse AAV ELISA Kits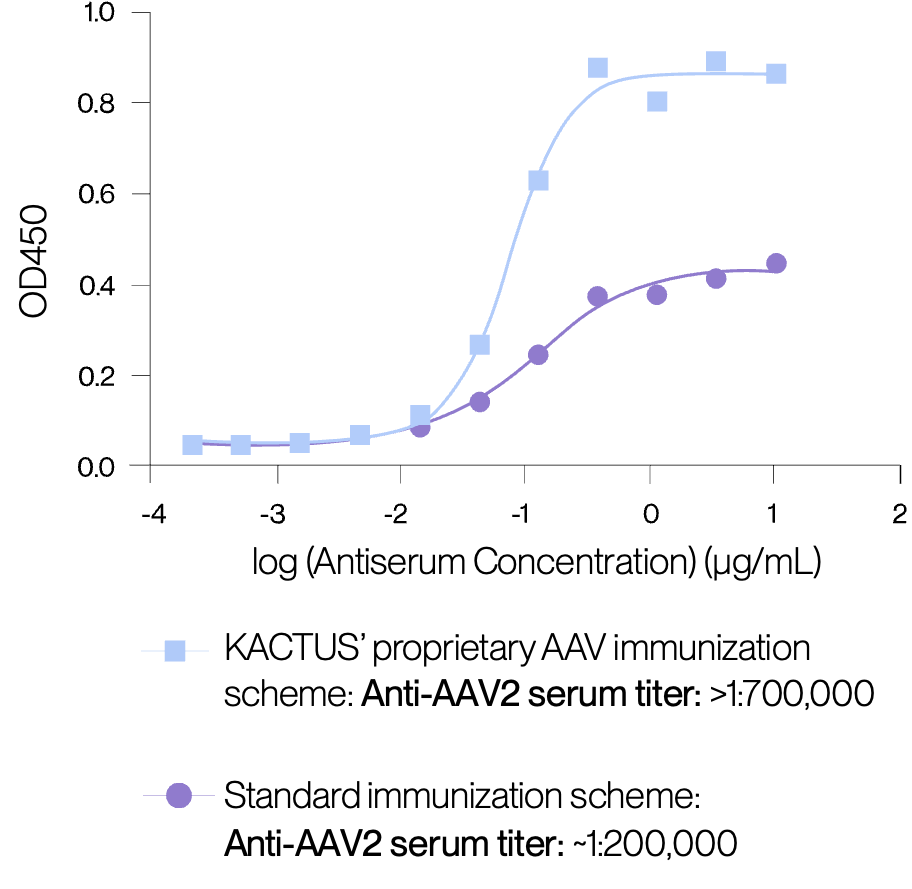
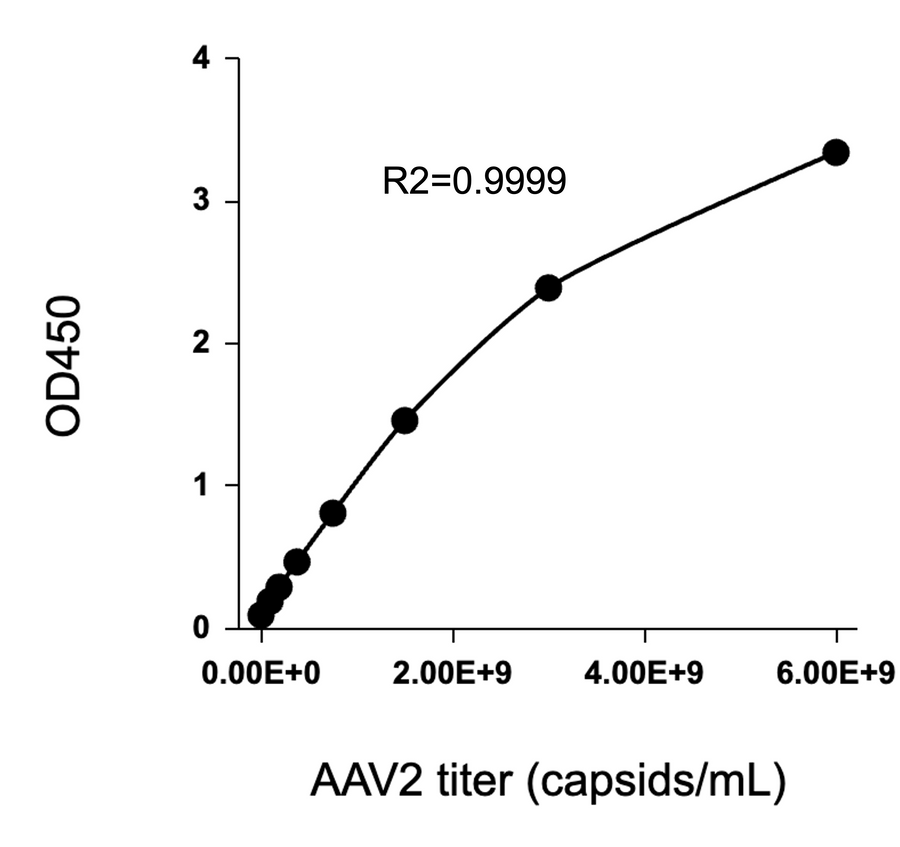
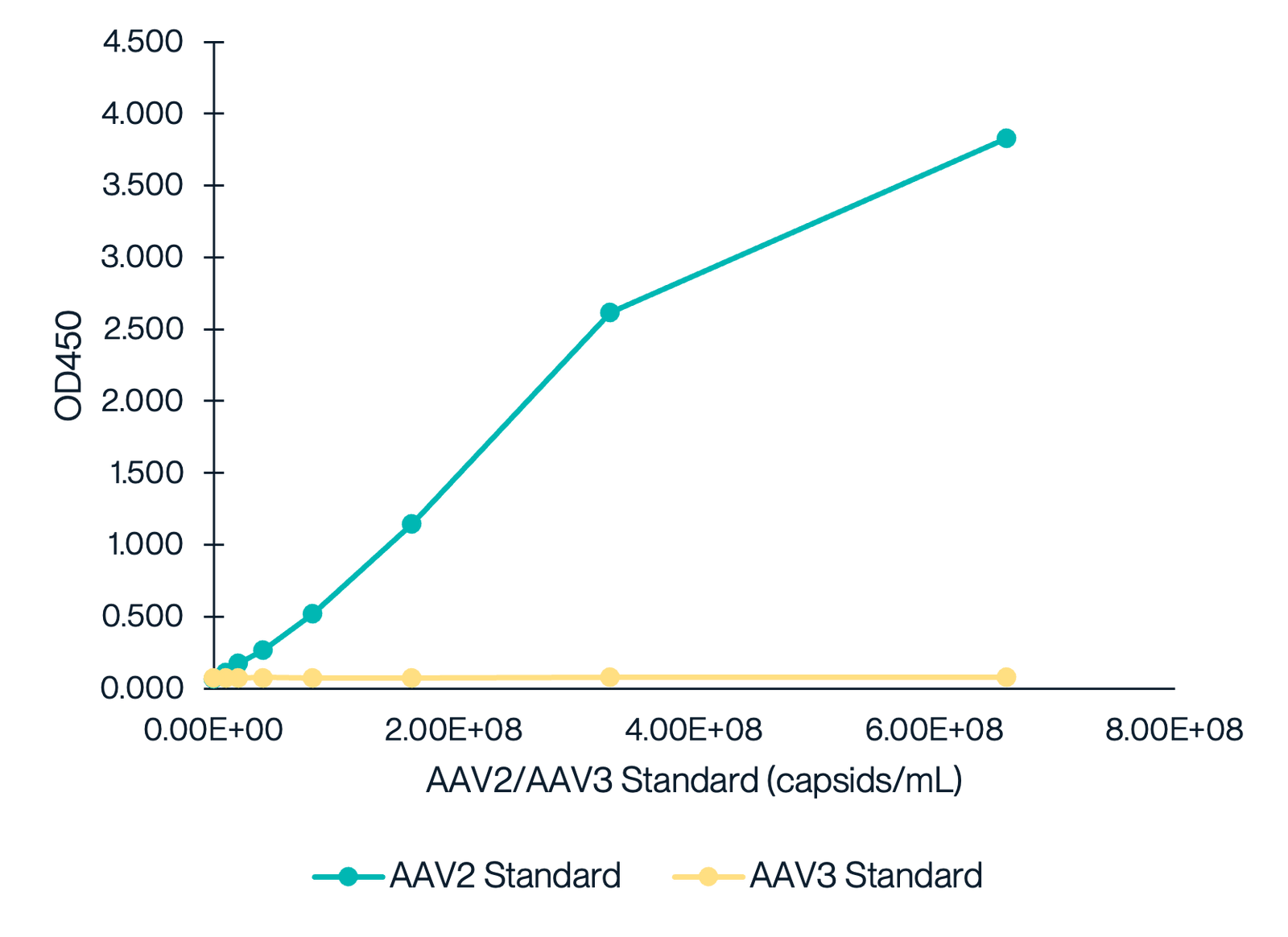
AAV2 Titration ELISA Kit Exhibits Near-Zero Cross-reactivity with AAV3
This graph demonstrates the cross-reactivity analysis of the AAV2 Titration ELISA Kit with AAV3. The data shows a dose-response curve for the AAV2 standard, while the AAV3 standard exhibits minimal reactivity, indicating the kit's high specificity for AAV2 with no cross-reactivity detected for AAV3.
Overview of AAV ELISA Kits
Sandwich ELISA assays are a trusted technique for quantification of adeno-associated virus (AAV) capsid proteins, enabling researchers to determine viral titers with high specificity and sensitivity. KACTUS has developed a portfolio of AAV Titration ELISA kits that are designed to detect and quantify AAV capsid proteins across different AAV serotypes. These kits are broadly applicable in gene therapy research.
Due to the low immunogenicity of AAVs, and its growing role in gene therapy, we have developed a proprietary AAV immunization scheme that produces high affinity anti-AAV monoclonal antibodies. We have extensively validated these ELISA assays to ensure that AAV titers can be accurately and consistently determined. Our AAV ELISA assays can be applied to both preclinical and clinical research, supporting vector production, potency testing, and the development of novel therapeutics. These kits offer high accuracy, reproducibility, and specificity for adeno-associated virus detection.
How the AAV ELISA Kits Work
The assay uses a sandwich ELISA method to analyze samples with varying AAV concentrations. Our kits have a broad linear range to detect viral capsids over a dynamic range. AAV particles are captured using serotype-specific monoclonal antibodies immobilized on an ELISA plate. A biotinylated detection antibody is added that binds to the capsid proteins. Following the addition of TMB, a substrate reaction generates an absorbance signal, which is subsequently analyzed against an 8-point standard curve to determine the concentration of viral particles.
This method is widely validated for research use, providing consistent and reproducible results for AAV vector characterization and quality control. The specificity of the ELISA kit allows for differentiation between intact AAV particles and denatured capsid proteins. The antibodies are also highly serotype-specific, for reliable quantification of specific AAVs.
Product Advantages
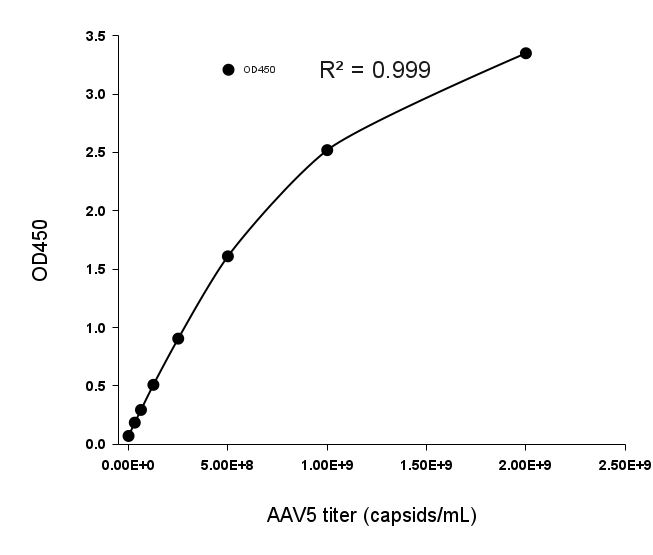
Wide Linear Range
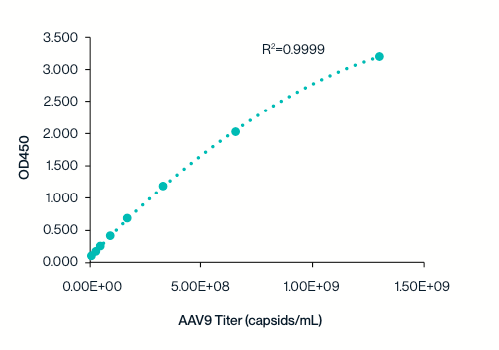
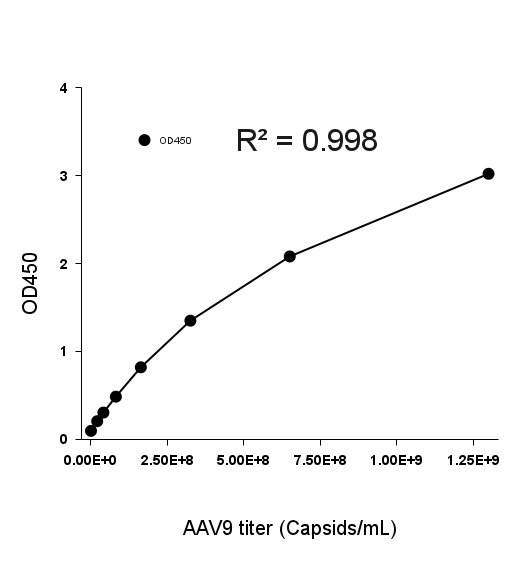
Example 8-point standard curve for the AAV9 Titration ELISA Kit.
The quantitative range of this kit is 2.03x107 - 1.30109 capsids/mL.
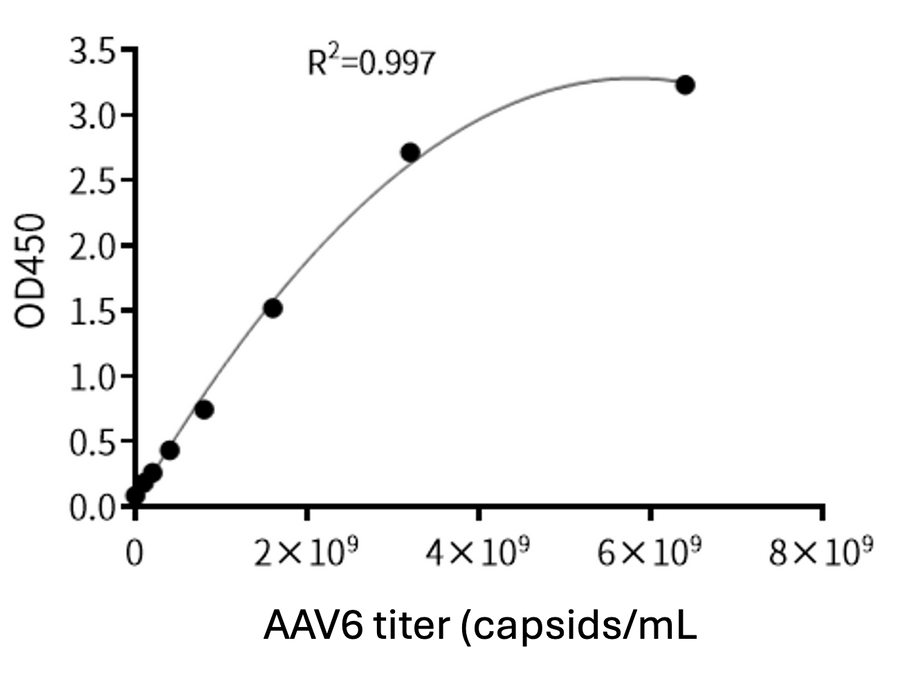
Batch Consistency
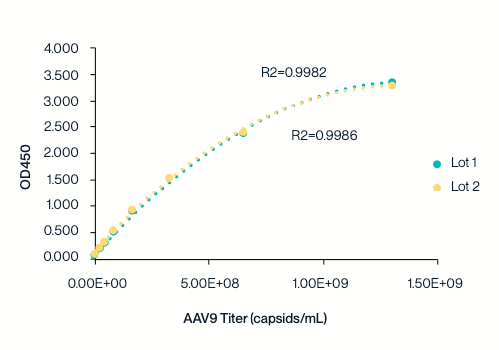
KACTUS AAV9 Titration ELISA kit standard curve tested across two batches.
The standard curve is highly reproducible and exhibits an R²>0.99.
Specificity to intact capsids
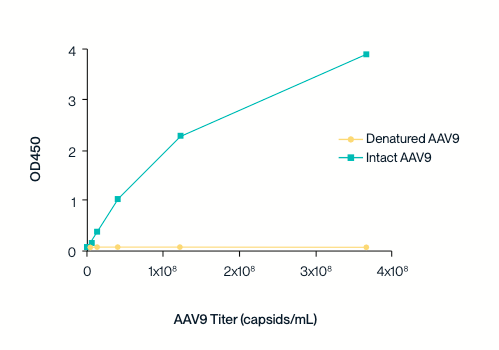
The ELISA sandwich assay was used to detect the binding of AAV9 antibodies to intact and denatured AAV9 capsid proteins.
The AAV9 detection antibody does not exhibit binding to denatured AAV9 capsid protein.
Specificity to individual serotypes
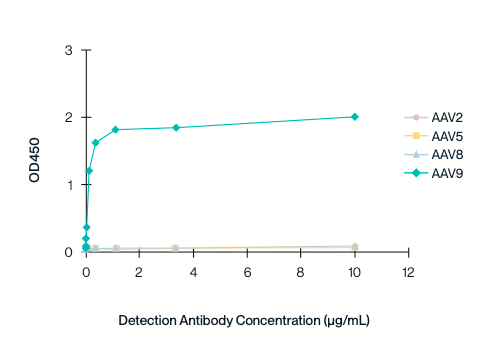
The cross-binding of different concentrations of AAV9 antibodies to capsid proteins of other serotypes (AAV2/5/8/9) was detected by indirect ELISA.
The AAV9 titration ELISA kit is highly specific to the AAV9 serotype and does not exhibit cross-reactivity with other AAV serotypes.
High Accuracy
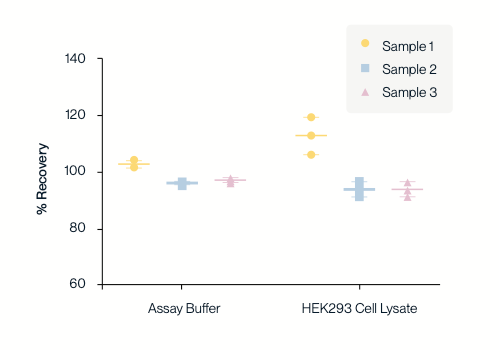
KACTUS AAV9 Titration ELISA kit was used to test the recovery rate of three AAV9 titers (high, medium, and low) in spiked dilution buffer (left) and HEK293 cell lysate (right).
The sample recovery rates of AAV9 samples with different titers detected in the dilution buffer and cell lysate were all between 80% and 120%, indicating that this kit has high accuracy in detecting AAV9 titers.
High Precision
| Sample | Runs | Average (capsids/mL) | Coefficient of Variation (%CV) |
|---|---|---|---|
| 6.5E+08 capsids/mL | n=10 | 6.18E+08 | 2.80% |
| 1.63E+08 capsids/mL | n=10 | 1.92E+08 | 1.90% |
| 2.03E+07 capsids/mL | n=10 | 2.17E+07 | 5.90% |
The same batch of AAV9 test kits were used to test three different titer AAV9 samples (high, medium and low). Each sample was tested 10 times. The average and coefficient of variation (%CV) of these 10 titer tests was calculated.
The %CV for all three sample concentrations (high, medium and low) was less than 10%, indicating that the AAV9 Titration ELISA kit has good intra-batch repeatability.
AAV Titration ELISA Kits FAQs
The kits quantify intact AAV capsid proteins using a sandwich ELISA format. Detection is specific to fully assembled capsids and does not include denatured or empty capsid fragments.
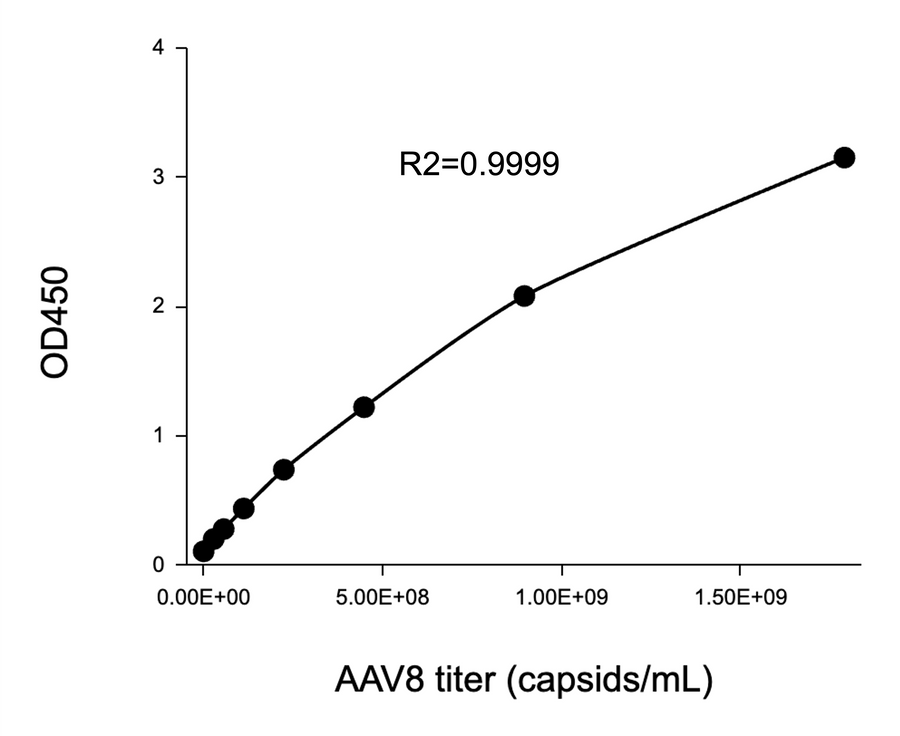
Yes. Each kit uses monoclonal antibodies with high specificity for a particular AAV serotype, such as AAV2, AAV5, AAV6, AAV8, or AAV9. Cross-reactivity with other serotypes is minimal to none.
No. The antibodies are specific to the native conformation of intact capsid proteins and do not bind denatured or degraded capsids.
Kits have a linear detection range typically spanning from approximately 2.03×10⁷ to 1.3×10⁹ capsids per mL, with high reproducibility across batches (R² > 0.99).
Each kit contains a pre-coated 96-well plate, AAV standard, capture and detection antibodies, streptavidin-HRP, assay buffer, wash buffer, TMB substrate, and stop solution.
Samples should be diluted in an assay buffer and tested in duplicate. A minimum of two dilutions per sample is recommended to ensure accuracy within the linear range of the standard curve.
No. Reagents from different batches should not be mixed, as this may affect the accuracy and reproducibility of results.
No. A fresh standard curve should be run on each plate to account for potential plate-to-plate variation and to ensure valid quantification.
Standard curve fitting may vary depending on lab software. Four-parameter logistic (4PL) is commonly used for ELISA data, but linear or polynomial models may be acceptable depending on the observed curve. Use the method that best fits your actual data.
No. The AAV Titration ELISA Kits are intended for research use only and are not approved for clinical diagnostics or therapeutic monitoring in humans.