Soluble TCR Expression & Affinity Analysis Services
Custom TCR Engineering for Immunotherapy Research
We're pleased to offer specialized custom services related to MHC and T cell receptor research, including the expression of Recombinant T cell receptors, and TCR-related drugs. Researchers often need soluble TCRs to study their role in antigen recognition, develop TCR-based therapies, and screen for therapeutic candidates targeting T cells in complex disease models.
In support of this, we offer custom expression services for Soluble TCR Monomers, fusion proteins, and specialized variants such as His-tagged TCRs. Our platform supports downstream applications such as CAR T cells development, adoptive cell therapy, and antibody-based T cell engagers. Additionally, we provide affinity analysis services including matured. T-cell receptor ELISAs and low affinity surface plasmon resonance (SPR) assays to assess interactions between TCR chains and peptide-loaded MHC complexes.

Production of various formats of Soluble TCR Monomers, including scFv-TCR, TCR-His, and Fc-fused variants designed for studies involving T cell cytotoxicity and peptide pulsing.
TCR engineering to optimize soluble secretion efficiency structural integrity, and pairing of TCR chains using computational modeling and structure-guided design.
SPR or ELISA analysis of soluble TCR & MHC interactions. Expression in systems like HEK293 cells and CHO-T cells, enabling high-yield recovery, appropriate folding, and post-translational modifications including potential glycosylation sites. Optional purification using Ni column or Protein A column, followed by quality screening for T-cell receptor molecules.
Request Quote:
We have successfully expressed a GP100. T cell antigen receptor fused with Anti-CD3, forming a Bispecific T Cell Engager that can simultaneously bind to HLA-A*02:01&B2M&GP100 (YLEPGPVTA) and CD3 heterodimers. This construct demonstrates high binding activity with potential applications in T-cell therapies and immune cells targeting.
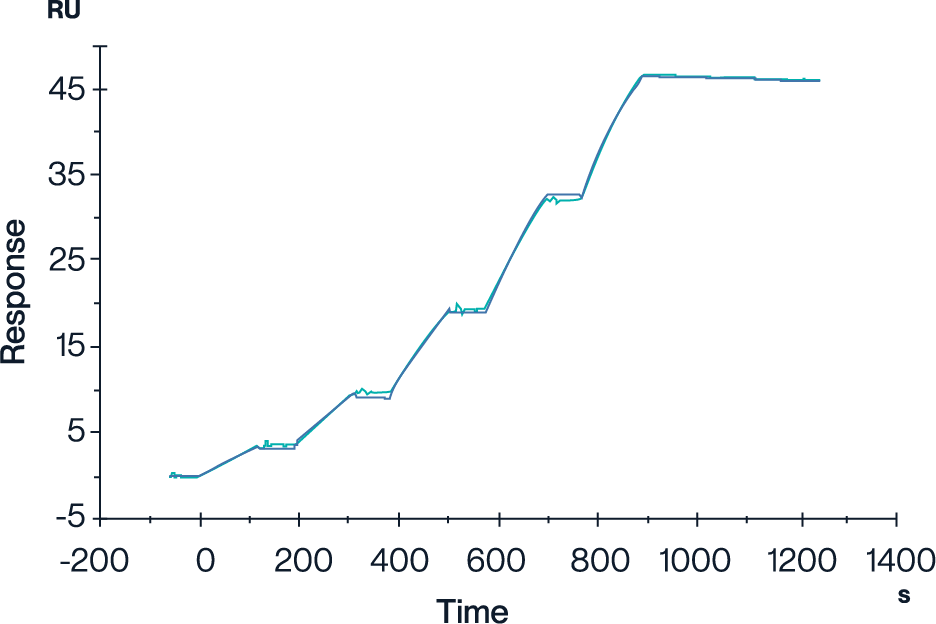
Figure 1. In surface plasmon resonance assay, the GP100 TCR & Anti-CD3 Bispecific Fusion can bind to the HLA-A*02:01&B2M&GP100 (YLEPGPVTA) complex tetramer, confirming antigen recognition capacity with an affinity constant of 0.196 nM.
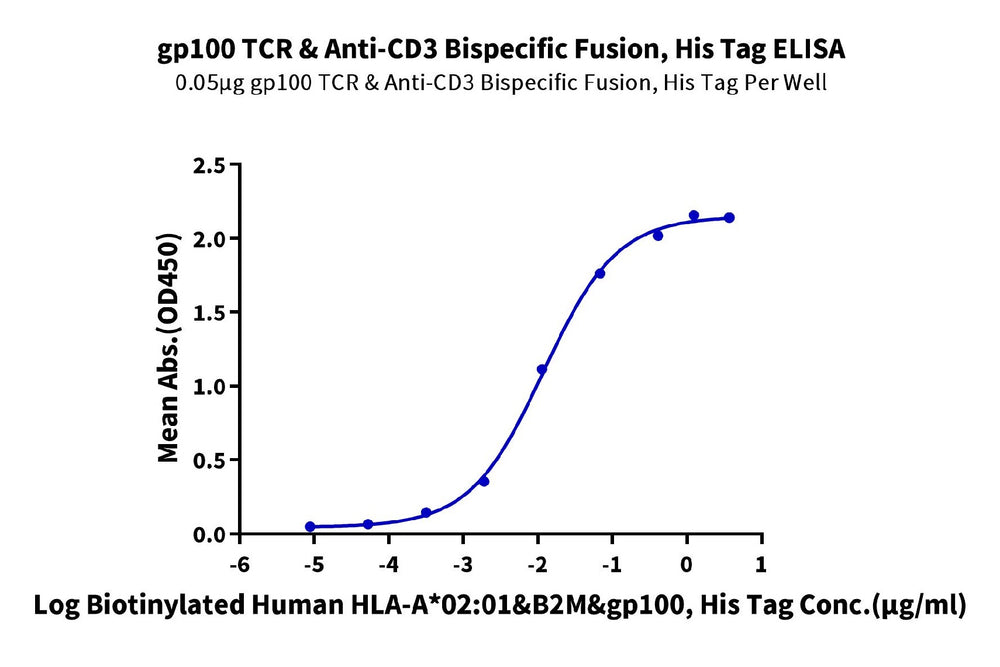
Figure 2. ELISA verification demonstrates that GP100 TCR & Anti-CD3 Bispecific Fusion can bind to HLA-A*02:01&B2M&GP100 (YLEPGPVTA) complex monomer with an EC50 of 11.8 ng/mL.
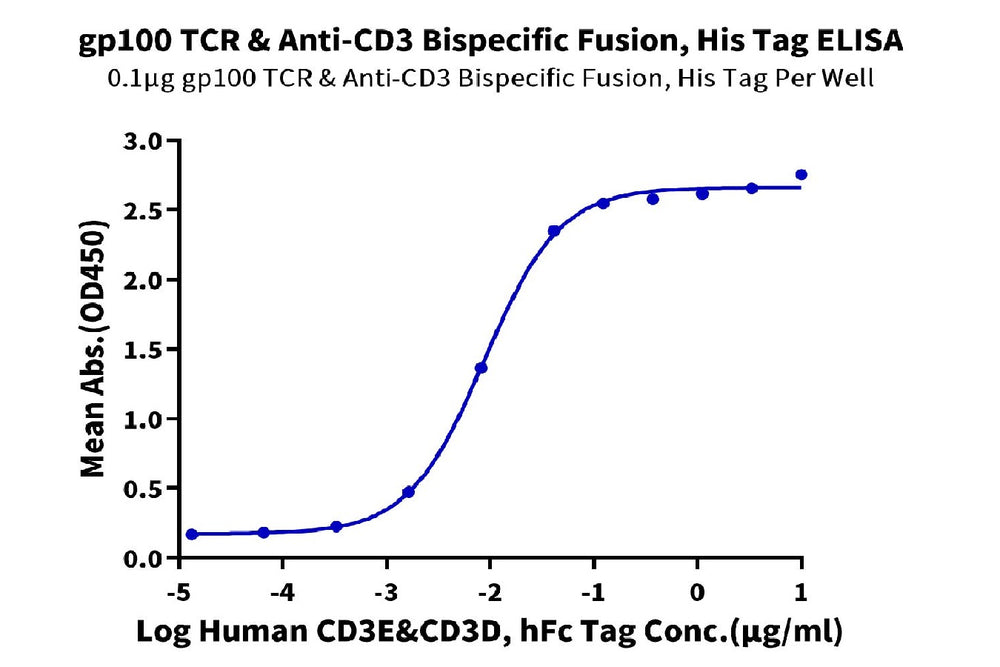
Figure 3. ELISA verification demonstrates that GP100 TCR & Anti-CD3 Bispecific Fusion can bind to Human CD3E&CD3D, with an EC50 value of 8.7 ng/mL.
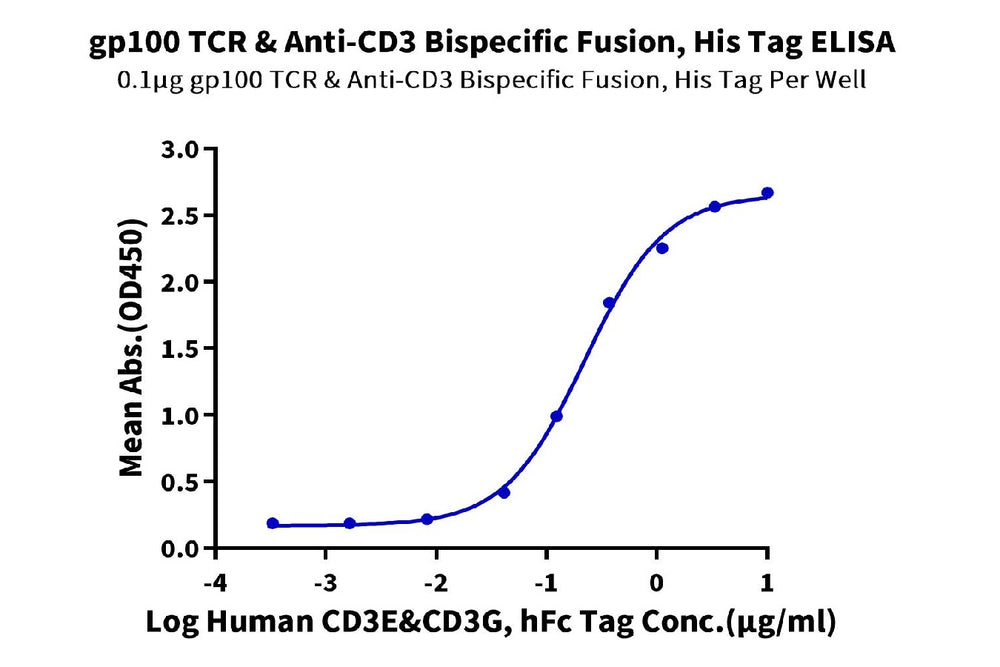
Figure 4. ELISA verification demonstrates that GP100 TCR & Anti-CD3 Bispecific Fusion can bind to Human CD3E&CD3G, with an EC50 value of 0.22 μg/mL.
We expressed an HLA-A*02:01&B2M&AFP (FMNKFIYEI) TCR which can successfully bind to our single-chain trimer MHC-peptide complex and our prMHC loaded with AFP peptide (FMNKFIYEI).
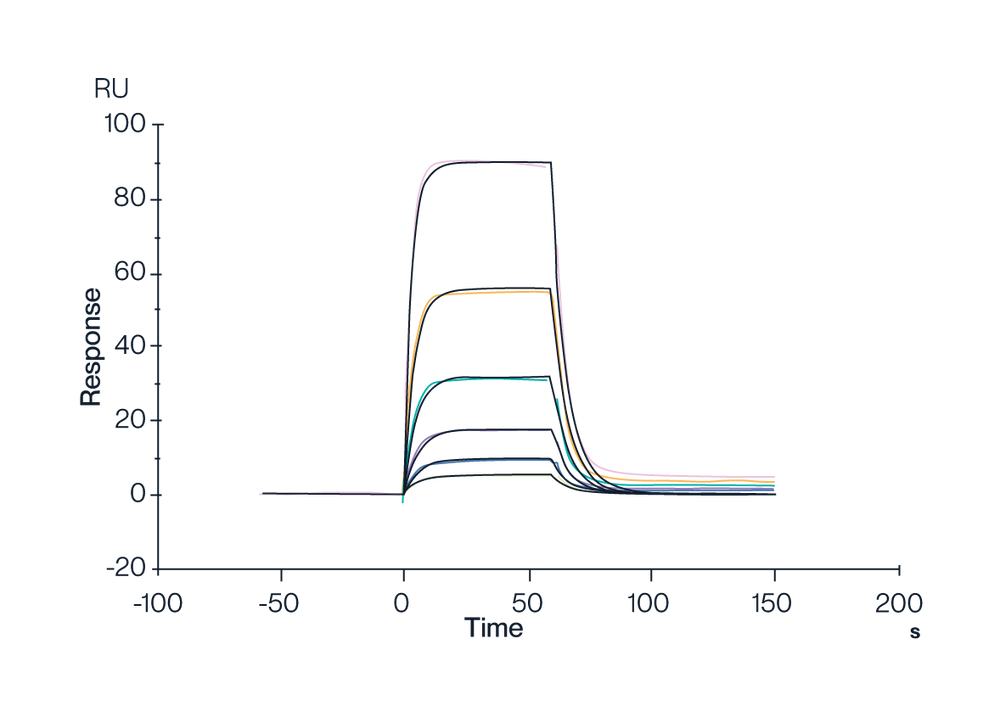
Figure 5. Single-chain trimer MHC, Human HLA-A*02:01&B2M&AFP (FMNKFIEl) Monomer, His-Avi Tag captured on CM5 Chip via Anti-His Antibody, can bind HLA-A*02:01&B2M&AFP (FMNKFIYEI) TCR with an affinity constant of 0.3991 µM as determined in SPR assay (Biacore T200). This expression was achieved using our mammalian system, with folding efficiency optimized through control of N-linked glycosylation enzymes.
Figure 6. Peptide-ready MHC, Human HLA-A*02:01&B2M, His-Avi Tag loaded with FMNKFIYEI peptide, captured on CM5 Chip via Anti-His Antibody, can bind HLA-A*02:01&B2M&AFP (FMNKFIYEI) TCR with an affinity constant of 0.326 µM as determined in SPR assay (Biacore T200), confirming stable interaction and supporting use in phage display or phage and yeast display platforms.
These studies also support workflows that involve T Cell Mate, LV TCR-T cells, 2A peptide incorporation strategies, or modeling T-cell receptor αβ diversity to improve binding precision and downstream therapeutic utility.
We manufacture a range of recombinant T cell receptor (rTCR) formats including Soluble TCR Monomers, scFv-TCRs, and Fc fusions. Our production capabilities support formats applicable in adoptive cell therapy, bispecific T cell engaging TCR research, and T-cell receptor αβ diversity profiling.
We apply computational and experimental strategies to optimize T cell receptor expression, pairing of TCR chains, and soluble secretion efficiency. Expression systems like CHO-T cells or HEK293 cells allow for improved folding and scalable yields for therapeutic development.
We provide high-precision analysis of T-cell receptor interactions with antigen-presenting cells using surface plasmon resonance or ELISA. This includes workflows involving rare TCRs like γσ T cells, pp65-specific T cells, or those involved in immunodominant CD8 T cell responses.
Our experienced team has engineered and tested hundreds of constructs targeting peptide presentation on MHC, including MUC1 peptide, immunodominant NLV peptide, and other tumor-associated epitopes displayed on CHO cell surface or tumor cell surface pHLAs.
Soluble TCRs are engineered, non-membrane-bound T cell receptors used for research or therapeutic applications. They mimic native antigen recognition but are produced as standalone proteins for easier handling in assays.
We support custom production of scFv-TCRs, TCR-His tags, TCR fusion proteins, and TCR-CD3 complexes, based on your project goals and downstream use (e.g., binding studies, bispecific constructs, etc.).
You’ll need to submit your TCR sequence, preferred expression format, any tags or fusion partners, and intended use (e.g., ELISA, SPR, functional assays). We’ll assess feasibility and provide a quote.
The standard timeline is 6–8 weeks including shipping, though complex constructs or additional validation (e.g., affinity testing) may extend this period slightly.
We primarily use mammalian systems (HEK293) for accurate folding and bioactivity. E. coli expression may be considered for certain constructs depending on size and complexity.
Yes. We perform SPR and ELISA-based affinity testing to measure TCR binding to specific MHC-peptide complexes. This helps confirm specificity and binding strength.
Yes. Our peptide-ready MHC monomers and tetramers allow you to load your peptide of interest and perform TCR binding assays independently.
All projects include purity and identity checks, which may include SDS-PAGE, Western blot, HPLC, ELISA, or SPR, depending on the intended use. Speak with our representatives to learn which QC will be included and which is available to request for an additional small fee.
Yes. Our team applies structure-based TCR engineering and modeling to improve expression yield, solubility, and functional conformation.
Yes. All client information, sequences, and project data remain strictly confidential. We support NDAs upon request and do not claim any rights to your intellectual property.