MaxNuclease™, GMP-Grade
Nonspecific endonuclease degrades all DNA & RNA for biologics manufacturing
With the rapid development of the biopharmaceutical field, the variety of biological products is increasing. At the same time, biological products have strict quality control systems, with residual nucleic acids being a key focus for regulatory agencies both domestically and internationally. Products such as AAV vectors, antibody drugs, vaccines, and recombinant protein drugs are produced using continuously passaged cells, and despite meticulous purification processes, residual host nucleic acids may still be present. These residual nucleic acids can pose risks such as pathogenicity and tumorigenicity. Hence regulatory agencies have strict guidelines for the amount of residual nucleic acids in biological products.
In addition, in the CAR-T cell therapy industry, large-scale purification of lentiviruses, protein extraction, and other fields also require strict control of nucleic acid residues. Currently, methods for removing nucleic acids mainly include enzymatic degradation, polyethyleneimine (PEI) precipitation, and ion exchange chromatography. Among these, enzymatic methods using nonspecific nucleases are highly favored for their efficiency, cost-effectiveness, and ease of use.
KACTUS has developed a GMP-grade universal nuclease—MaxNuclease™, which degrades all DNA and RNA into 2-5 base oligonucleotides. It has been developed and validated according to GMP manufacturing standards to ensure the traceability of raw materials throughout the production process. This product has been registered with the U.S. FDA Drug Master Files (#036799), providing documentation on aspects such as production processes and control, and material control.
What is MaxNuclease™?
MaxNuclease™ is identified from Serratia marcescens and is genetically engineered and expressed in E. coli under cGMP manufacturing standards. MaxNuclease™ is a non-specific nuclease with high activity and specificity that degrades all forms of nucleic acids including single- and double-stranded, linear and circular nucleic acids.
How does MaxNuclease™ work?
MaxNuclease™ is a homodimer of two 30 kDa subunits containing two disulfide bonds that are essential for activity and stability. It hydrolyzes internal phosphodiester bonds between nucleotides in nucleic acids to produce 5'-monophosphate oligonucleotides of 2-5 bases in length.
What is MaxNuclease™ good for?
Products such as lentiviral or AAV vectors, antibody drugs, vaccines, and recombinant protein drugs are expressed and produced by continuously passaged cells. Even after a fine purification process, host nucleic acids may remain in the products, and the residual nucleic acids may cause pathogenicity, tumorigenesis, etc. Residual nucleic acids need to be removed to a safe level in the final drug product.
Manufactured in a GMP-compliant facility

Raw materials free from animal-derived components
Low Endotoxin: ≤ 0.01 EU/kU

Strict quality management for clinical manufacturing
FDA Drug Master Files: DMF #036799
| Parameter | Specification |
|---|---|
| Catalog No. | GMP-NUC-SE101 |
| FDA Drug Master Files | #36799 |
| Source | E. coli with endonuclease gene from Serratia marcescens |
| Molecular Weight | Approximately 27.8 kDa |
| Formulation | 20mM Tris-HCl, 20mM NaCl, 2mM MgCl2, 50% Glycerol, pH 8.0 |
| Storage | Store at -20±5°C. Avoid repeated freeze-thaw. |
| Activity | ≥250 U/µL analyzed by degradation of Herring Sperm DNA |
| Unit Definition | One unit corresponds to the amount of enzyme required to produce a change in absorbance at 260 nm of 1.0 in 30 minutes, at 37°C and pH 8.0. |
| Assay | Specification |
|---|---|
| Activity (Dissolve herring sperm DNA) | ≥ 250 U/µL |
| Purity (Bis-Tris) | ≥ 95% |
| Purity (SEC-HPLC) | ≥ 99% |
| Residual Protease | Negative |
| Residual Host Protein | ≤ 10 ppm |
| Endotoxin | ≤ 0.01 EU/kU |
| Sterility | Negative |
| Residual Heavy Metal | ≤ 10 ppm |
| Mycoplasma | Negative |
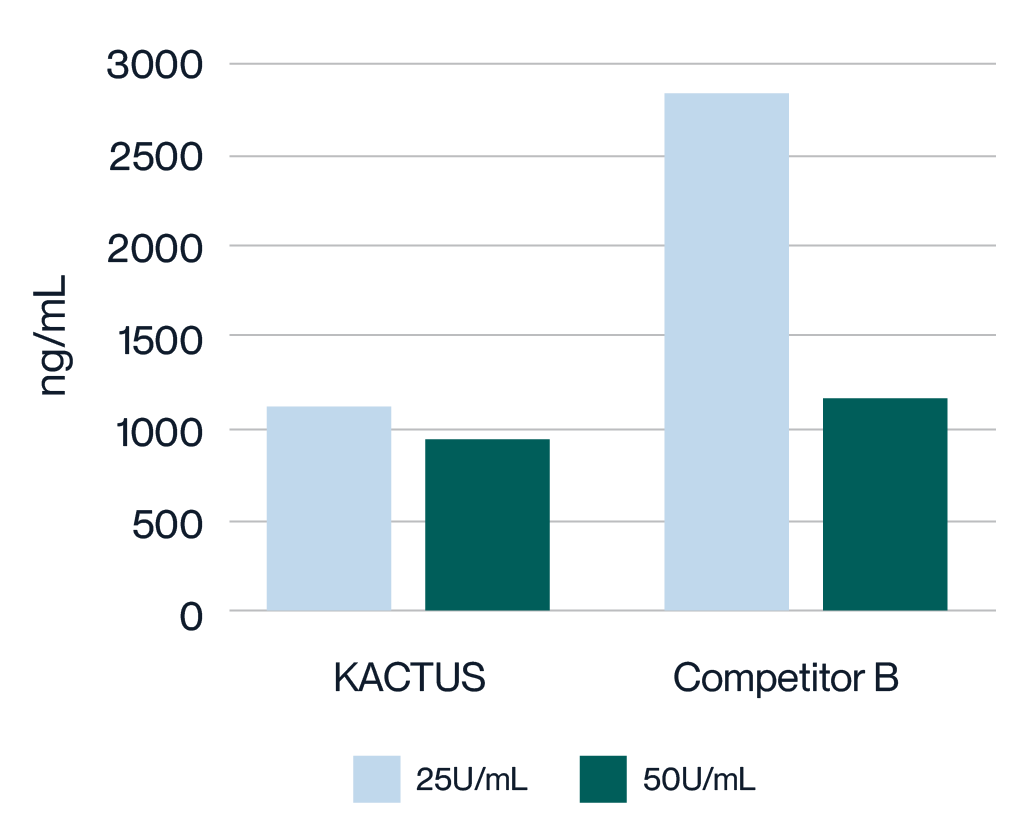
Virus harvest solution was treated with 25U/mL and 50U/mL endonuclease at 37°C for 2 hours, respectively. Detection of Host Cell DNA (HCD) residue was analyzed. MaxNuclease™ has higher degradation activity versus Competitor B demonstrated by lower HCD residue for both 25U/mL and 50U/mL working concentrations.
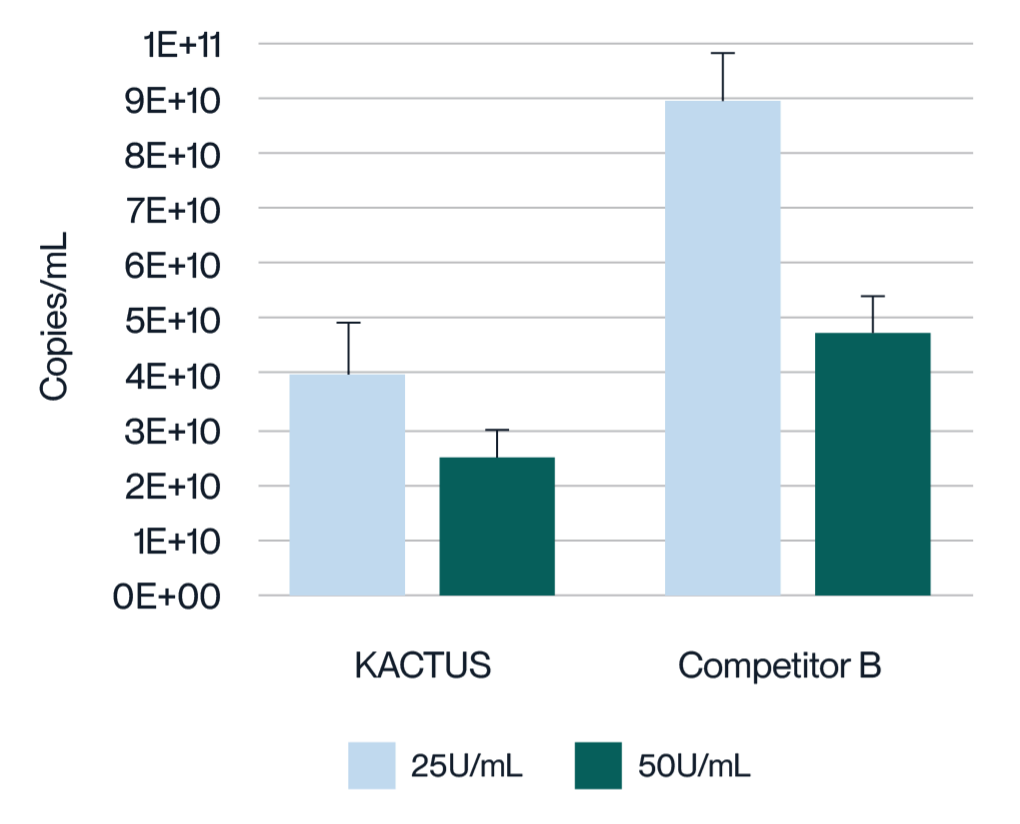
Virus harvest solution was treated with 25U/mL and 50U/mL endonuclease at 37°C for 2 hours, respectively. Detection of plasmid DNA (pDNA) residue was analyzed. MaxNuclease™ has higher degradation activity versus Competitor B demonstrated by lower pDNA residue for both 25U/mL and 50U/mL working concentrations.

MaxNuclease™ added to PCR product, genomic DNA, and plasmid DNA shows comparable degradation activity of nucleic acids to leading competitors.
Our MaxNuclease™ has been developed and verified with reference to cGMP production standards. The manufacturing process is in accordance with GMP production standards to ensure the traceability of raw materials in the production process. At present, this product has also been filed with the U.S. FDA Drug Master Files (DMF #036799).
MaxNuclease™ is available in long-term bulk supply with batch-to-batch consistency to ensure suitability for industrial applications.
→ ISO13485 Accreditation
→ Digital Manufacturing Execution System (MES)
→ Process and Analytical method validation
→ Batch-to-batch stability and consistency
→ Free from antibiotic residues and raw materials of animal origin
→ Comprehensive records for batch production
→ Pharmaceutical Class A & C Clean Room
→ Validated and maintained equipment
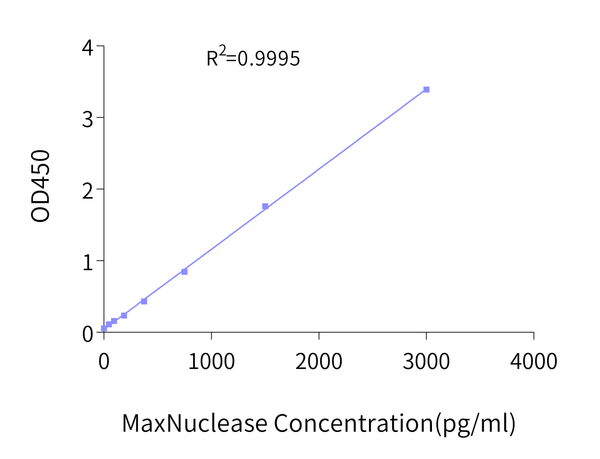
After nucleic acid removal using MaxNuclease™, the residual enzyme in the product or sample must be evaluated. For residual MaxNuclease™ detection, KACTUS has developed a highly sensitive and specific sandwich ELISA quantification kit, with a sensitivity of up to 23 pg/mL.
MaxNuclease™ is a non-specific endonuclease derived from Serratia marcescens. It is produced in E. coli and purified under GMP-compliant conditions. The enzyme degrades both DNA and RNA into short oligonucleotides (2–5 bases), supporting residual nucleic acid removal in biomanufacturing.
MaxNuclease™ degrades all forms of nucleic acids including single-stranded and double-stranded DNA and RNA, whether linear, circular, plasmid, genomic, or PCR-amplified.
One unit of activity is defined as the amount of enzyme required to produce a change in absorbance of 1.0 at 260 nm in 30 minutes at 37°C using herring sperm DNA. MaxNuclease™ is verified to have at least 250 U/µL.
MaxNuclease™ requires 1 to 2 mM Mg²⁺ for activity. It performs best at 37°C and pH 8.0. Activity decreases at lower temperatures and is sensitive to inhibitors such as EDTA and high concentrations of monovalent salts.
It is commonly used for removing residual nucleic acids from AAV or lentiviral vectors, protein biologics, vaccines, and CAR-T manufacturing workflows. It also reduces lysate viscosity and improves purification efficiency.
MaxNuclease™ is typically added after cell harvest and before purification. A typical working concentration ranges from 25 to 50 U/mL, depending on the buffer system, temperature, and target nucleic acid load.
Stored at –20°C, the enzyme remains stable for at least three years. It retains more than 90 percent of activity after 7 days at 25°C and 37°C when kept in a suitable buffer.
MaxNuclease™ is supplied in 20 mM Tris-HCl, 20 mM NaCl, 2 mM MgCl₂, and 50 percent glycerol at pH 8.0. It should be stored at –20°C and protected from repeated freeze-thaw cycles.
It can be removed through common downstream purification methods such as ion exchange chromatography, depth filtration, or tangential flow filtration. Residual enzymes can be quantified using our MaxNuclease ELISA kit.
Each lot is tested for activity, purity by SDS-PAGE and SEC-HPLC, and residual contaminants including host proteins, proteases, heavy metals, endotoxins, and mycoplasma. Endotoxin content is confirmed to be ≤ 0.01 EU/kU.