Recombinant KLK Proteins (Kallikrein Proteases)
The Kallikrein-related peptidase (KLK) family is comprised of 15 secreted serine proteases (KLK1–KLK15), synthesized as inactive zymogens (pro-forms). Upon cleavage of their N-terminal pro-peptide, KLKs adopt an active conformation and precisely cleave peptide substrates to regulate proteolytic cascades essential for tissue homeostasis.
KLK - A Hot Target in Disease
Dysregulation of KLK expression or activity is strongly linked to multiple pathologies including:
- Cardiovascular Disease
- Cancer (Prostate, Ovarian, Breast)
- Skin Disorders (e.g. Atopic Dermatitis, Netherton syndrome, etc.)
- Neurodegenerative Diseases (e.g. Alzheimer's Disease, Multiple Sclerosis, etc.)
- Airway Diseases (e.g. asthma, chronic bronchitis)
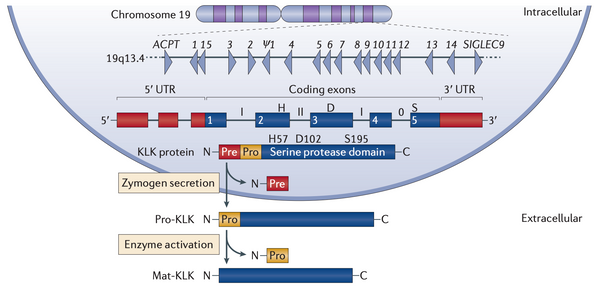
KLK genes are expressed as enzymatically inhibited zymogens
https://doi.org/10.1038/nrd4534
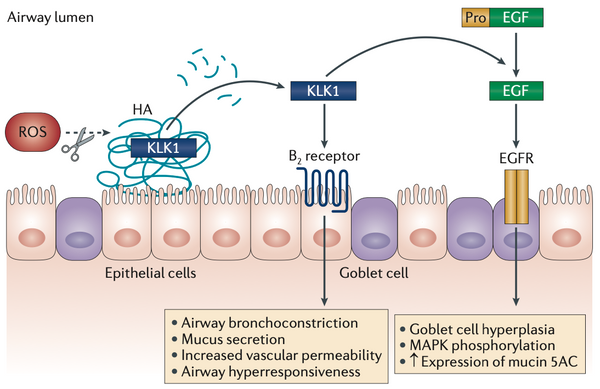
Pathological activity of KLK contributes to progression of airway diseases
Comprehensive Collection of KLK Enzymes
We offer most KLKs in both pro-form and enzymatically active forms (with more coming soon!), enabling simpler processing and faster functional studies.
Product Applications:
Drug & Antibody discovery:
Immunization
In Vitro Antibody/Inhibitor Screening
Antibody Affinity & Epitope Binning
Structural Characterization
Interaction & Kinetic Analysis
Binding Kinetics - SPR / BLI
Quantitative Immunoassays - ELISA
Cell-based Functional Assays
Product Features:
→ High protein binding affinity
→ Enzymatic activity data
→ Ultra-low endotoxin (<0.01 EU/μg) - critical for cell-based assays and in vivo studies
→ Mammalian Expression
→ Various tags (His, Flag, Biotin, etc.)
→ Multi-Species options, ideal for cross-species validation
Product Validation
KLK5: Regulator of Skin Homeostasis
KLK5 is a key serine protease primarily expressed in the epidermis, where it serves as a master initiator of the enzymatic cascade that regulates skin desquamation. By activating secondary proteases like KLK7, it maintains the delicate balance of the skin barrier. Its dysregulation is a central driver in inflammatory conditions such as Netherton syndrome, atopic dermatitis, and rosacea.
See our blog on the therapeutic landscape of KLK5/7 in these skin diseases →
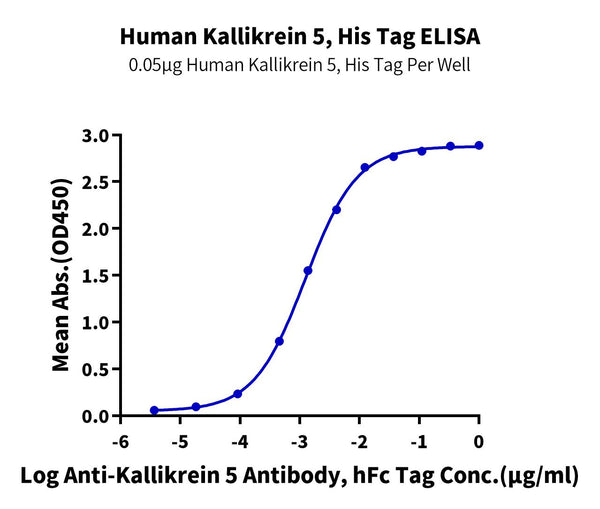
Immobilized Human Kallikrein 5, His Tag at 0.5ug/ml (100ul/Well) on the plate. Dose response curve for Anti-Kallikrein 5 Antibody, hFc Tag with the EC50 of 3.7ng/ml determined by ELISA (QC Test).
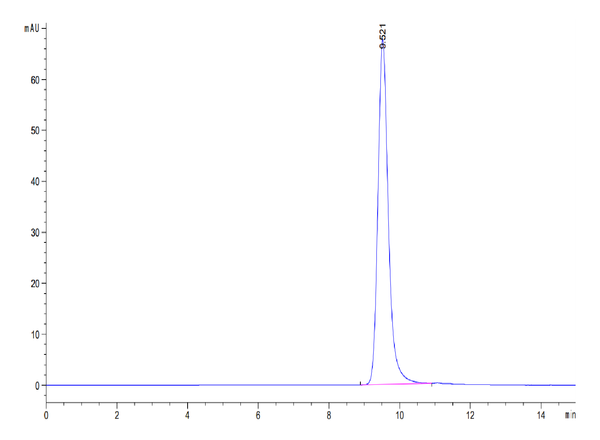
The purity of Human Kallikrein 5 is greater than 95% as determined by SEC-HPLC.
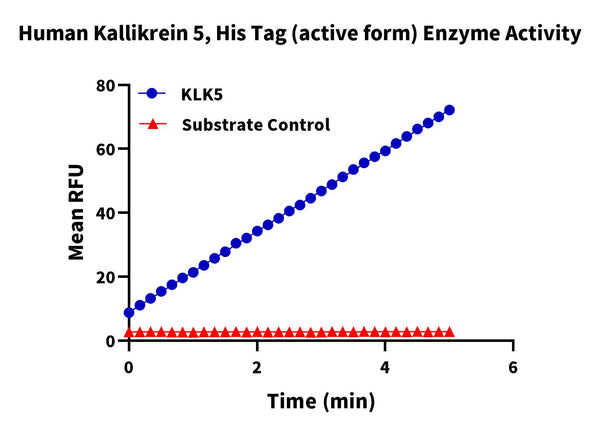
Measured by its ability to cleave the fluorogenic peptide substrate Boc-VPR-AMC. The specific activity is >400 pmol/min/µg (QC Test).
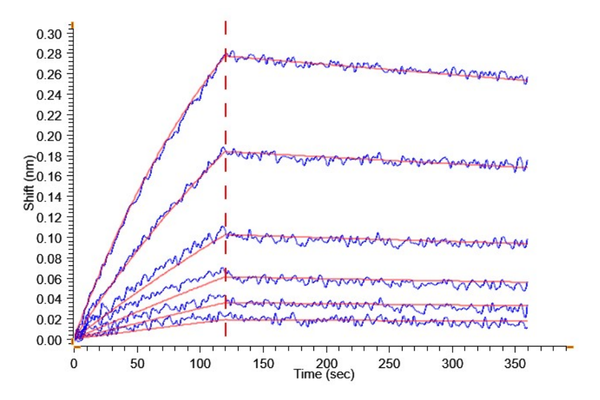
Loaded Anti-Kallikrein 5 Antibody, hFc Tag on ProA-Biosensor can bind Human Kallikrein 5, His Tag with an affinity constant of 1.23 nM as determined in BLI assay (Gator® Prime).
KLK2: New Target for Prostate Cancer
KLK2 (hK2) is a highly prostate-specific serine protease that shares 80% amino acid homology with PSA (KLK3) and acts as its primary physiological activator. Regulated by androgen receptor signaling, it is overexpressed throughout the prostate cancer disease continuum (including metastatic castration-resistant stages) where it facilitates tumor growth, migration, and angiogenesis. Beyond its role as a diagnostic biomarker, the recent discovery of its cell surface expression has established KLK2 as a promising therapeutic target for radioligand and CAR T-cell therapies.
See our blog on the latest clinical developments of KLK2 drugs in Prostate Cancer →
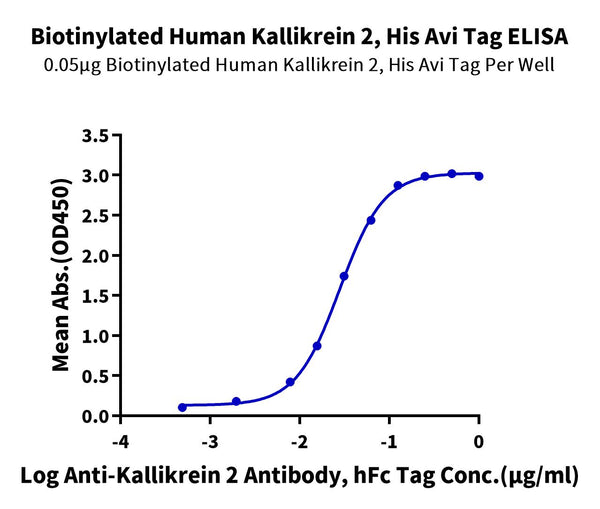
Immobilized Biotinylated Human Kallikrein 2, His Avi Tag at 0.5ug/ml (100ul/well) on the streptavidin precoated plate (5ug/ml). Dose response curve for Anti-Kallikrein 2 Antibody, hFc Tag with the EC50 of 28.0ng/ml determined by ELISA.
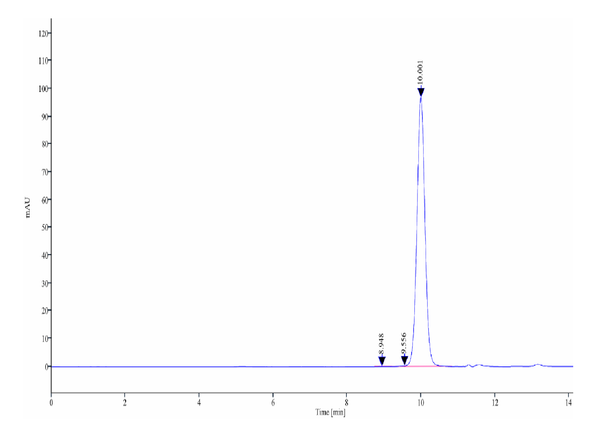
The purity of Biotinylated Human Kallikrein 2 is greater than 95% as determined by SEC-HPLC.
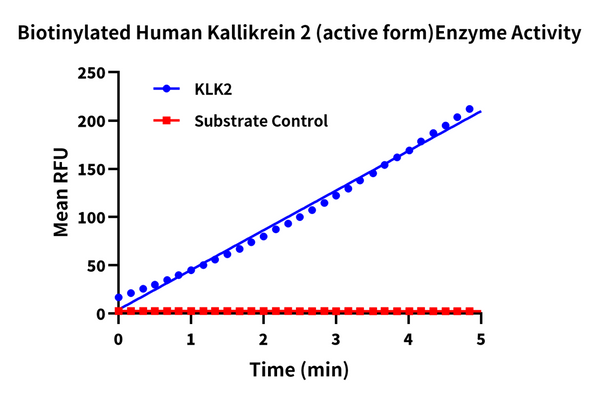
Measured by its ability to cleave the fluorogenic peptide substrate Boc-VPR-AMC. The specific activity is >400 pmol/min/µg (QC Test).
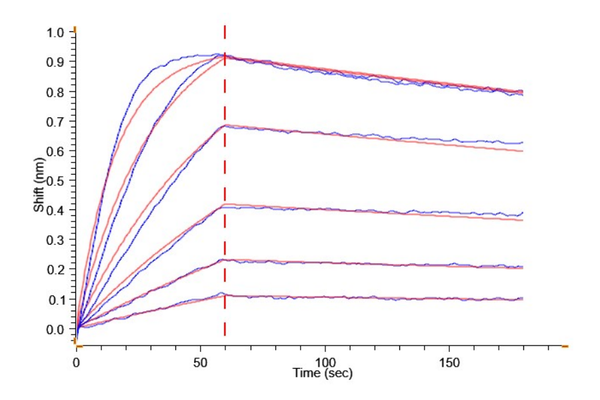
Loaded Anti-Kallikrein 2 Antibody, hFc Tag on ProA-Biosensor can bind Biotinylated Human Kallikrein 2, His-Avi Tag with an affinity constant of 1.89 nM as determined in BLI assay (Gator® Prime).
Resources
Check out our blogs and informational content for a deeper dive into KLK enzyme proteins: