About MHC Proteins
Major Histocompatibility Complex (MHC) is a highly polymorphic family of cell surface proteins, also known as HLA in humans. MHC binds to peptides derived from degraded intracellular antigens to form MHC-peptide complexes, which are then transported to the cell surface. These MHC complexes are recognized by corresponding T cell receptors (TCR) to initiate an immune response. In humans, the MHC complexes involved in antigen presentation are mainly MHC class I and MHC class II. MHC class I, when bound to antigenic peptides, is recognized by CD8+ T cells, while MHC class II, when bound to antigenic peptides, is recognized by CD4+ T cells. Targeting MHC complexes can achieve the killing of tumor cells.
KACTUS has successfully developed a series of MHC complexes, including monomers, tetramers, chimeric MHCs, and peptide-ready MHCs, as well as related custom services, fully supporting the development of tumor immunotherapy drugs. Our MHC complex portfolio covers popular targets such as NY-ESO-1, KRAS, AFP, and HPV16 which can be used to meet the diverse needs of immunology, molecular screening, TCR affinity validation, blocking assays, and other applications.
Product Features

Catalog Products
Our extensive catalog includes a wide range of MHC monomers and tetramers that are readily available to meet your research needs. Whether you are studying antigen presentation or T cell responses, our products are designed to provide reliable and consistent results.

Choice of Allele
We offer a diverse selection of alleles to ensure you can find the specific MHC molecules required for your studies. Our alleles cover various species and haplotypes, enabling precise and comprehensive immunological research.
Verified Bioactivity
Each of our MHC monomers and tetramers undergoes rigorous testing to verify bioactivity. This ensures that our products will perform as expected in your assays, providing you with confidence in your results.

Biotinylation & Fluorescent Labels
Enhance your experiments with our biotinylated and fluorescently labeled MHC monomers and tetramers. These options provide flexibility in detection methods, allowing you to choose the best approach for your flow cytometry, microscopy, or other analytical techniques.
Browse by Allele
MHC complexes play a crucial role in the immune system by presenting antigenic peptides to cytotoxic CD8+ T cells (class I) or helper CD4+ T Cells (class II).
MHC I are heterotrimers consisting of a transmembrane MHC heavy chain, a light chain known as β2-microglobulin (β2m), and an 8-10 peptide antigen. The heavy chain contains two peptide binding domains (α1 and α2), an immunoglobulin-like domain (α3), and a transmembrane region. The folding of the α1 and α2 domains forms a groove where peptide antigens bind to the MHC I complex. β2m stabilizes the peptide binding groove and MHC I presentation.
Class I Alleles - Human
Other Alleles
For other class I or non-classical alleles, try our custom MHC services.
Browse by Antigenic Peptide
MHC binds peptide fragments of intracellular antigens to form MHC-peptide complexes, which are transported to the cell surface and recognized by T cell receptors (TCRs) to initiate an immune response. Each nucleated cell expresses 5x10⁵ copies of each MHC I, presenting various peptides simultaneously to cytotoxic T cells. Their unique binding with TCRs is crucial for adaptive immunity and TCR-related therapies like TCR-T development. Genomic mutations in cancers produce tumor-specific peptide antigens (neoantigens), presented by MHC, which enable the immune system to target cancer cells.
Custom Peptide
For MHCs with custom peptide sequences, submit our Custom MHC Request Form.
Alternatively, try a Peptide-Ready MHC to load your peptide in-house onto an MHC tetramer or monomer.
Product Validation Data
We've demonstrated via ELISA and SPR binding assays that our MHC-peptide complexes and loaded peptide-ready MHCs have good binding affinity to TCRs. Our MHC products are well-suited for applications such as antibody binding, affinity research, TCR screening, and blocking assays.
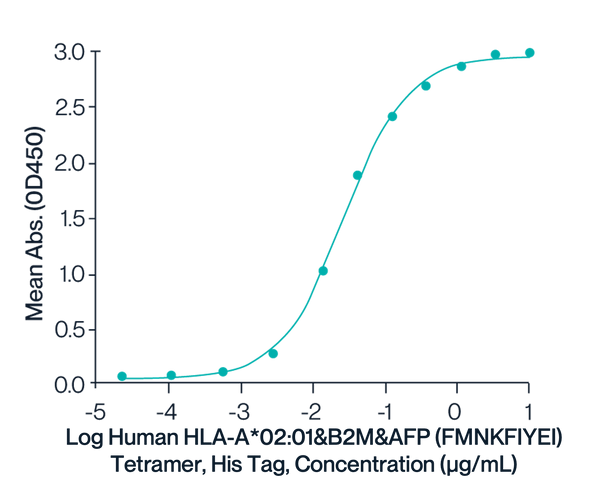
HLA-A*02:01 Tetramer & FMNKIYEI
Figure 1. Immobilized HLA-A*02:01&B2M&AFP (FMNKFIYEI) TCR at 2µg/ml (100ul/well) on the plate can bind HLA-A*02:01&B2M&AFP (FMNKFIYEI) Tetramer, His Tag with an EC50 of 26.6ng/ml determined by ELISA.
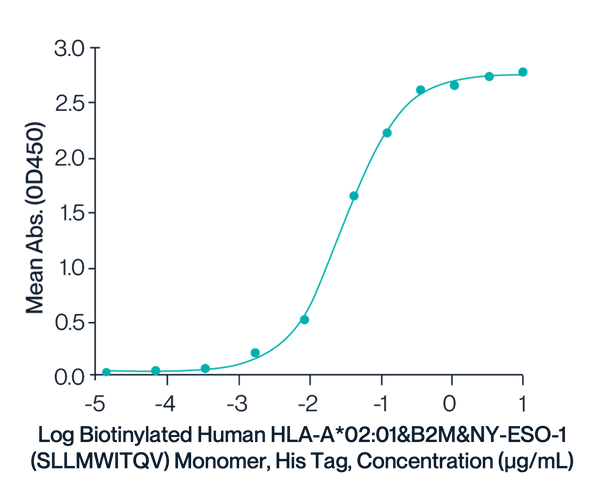
Biotinylated HLA-A*02:01 Monomer & SLLMWITQV
Figure 2. Immobilized Anti-HLA-A*02:01&B2M&NY-ESO-1 (SLLMWITQV) Antibody, hFc Tag at 1ug/ml (100ul/well) on the plate. Dose response curve for Biotinylated Human HLA-A*02:01&B2M&NY-ESO-1 (SLLMWITQV) Monomer, His Tag with the EC50 of 31.3ng/ml determined by ELISA.
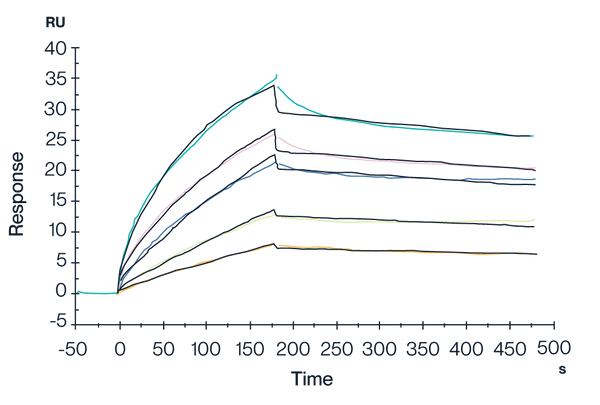
HLA-G Tetramer & RIIPRHLQL
Figure 3. Human LILRB2, hFc Tag captured on CM5 Chip via Protein A can bind Human HLA-G&B2M&Peptide (RIIPRHLQL) Tetramer with an affinity constant of 4.62 nM as determined in SPR assay (Biacore T200).
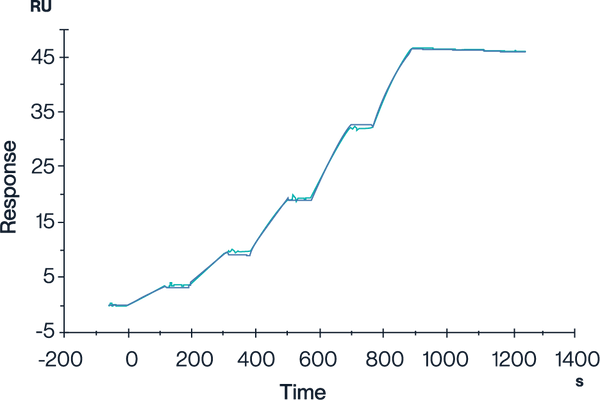
HLA-A*02:01 Tetramer & YLEPGPVTA
Figure 4. In SPR analysis, the GP100 TCR & Anti-CD3 Bispecific Fusion can bind to the HLA-A*02:01&B2M&GP100 (YLEPGPVTA) complex tetramer, with an affinity constant of 0.196 nM.
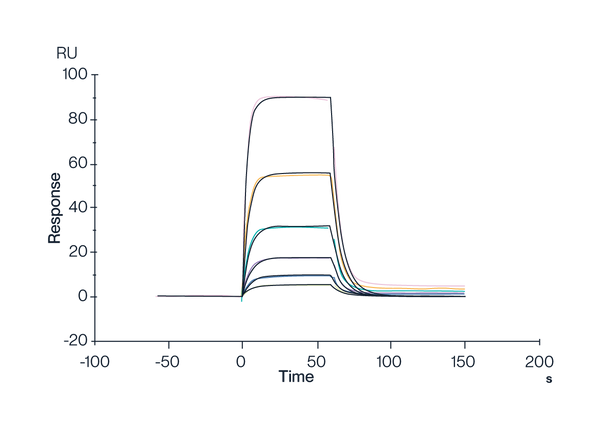
HLA-A*02:01 Monomer & FMNKFIYEI
Figure 5. Human HLA-A*02:01&B2M&AFP (FMNKFIEl) Monomer, His Tag captured on CM5 Chip via Anti-His Antibody can bind HLA-A*02:01&B2M&AFP (FMNKFIYEI) TCR with an affinity constant of 0.3991 µM as determined in SPR assay (Biacore T200).
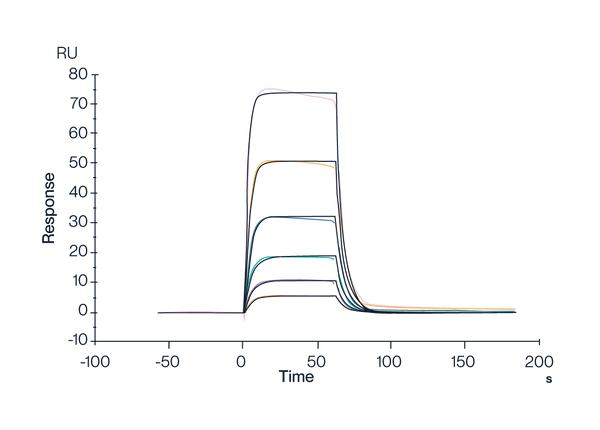
HLA-A*02:01 Tetramer & FMNKFIYEI
Figure 6. Human HLA-A*02:01&B2M&AFP (FMNKFIEl) Tetramer, His Tag captured on CM5 Chip via Anti-His Antibody can bind HLA-A*02:01&B2M&AFP (FMNKFIYEI) TCR with an affinity constant of 0.111 µM as determined in SPR assay (Biacore T200).
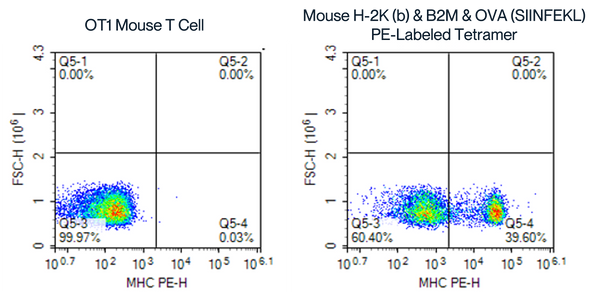
PE-SA Assembled Tetramer with SIINFEKL, OT1 Mouse Whole Blood
Figure 7. Flow Cytometry Analysis of OT1 Mouse Whole Blood Cells and T Cells Using PE-SA Assembled Tetramers. The right panel displays the binding of OT1 mouse T cells to the Mouse H-2K(b) & B2M & OVA (SIINFEKL) PE-SA assembled tetramer, with a significant shift in fluorescence and higher percentage of MHC PE-H positive cells (39.60% in Q5-4), compared to the left control, demonstrating effective binding and tetramer assembly.
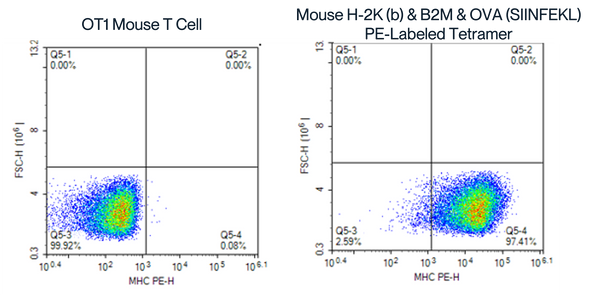
PE-SA Assembled Tetramer with SIINFEKL, Peptide-Pulsed OT1 Mouse Spleen Blood
Figure 8. Flow Cytometry Analysis of SIINFEKL Peptide-Pulsed OT1 Mouse Spleen Cells Using PE-SA Assembled Tetramers. The right panel displays the binding of OT1 mouse spleen cells to the custom-assembled tetramer (Mouse H-2K(b) & B2M & OVA (SIINFEKL) PE-SA), with a significant shift in fluorescence and higher percentage of MHC PE-H positive cells (97.41% in Q5-4), demonstrating effective binding and tetramer assembly.
Available Products
Browse our full catalog of 200+ major histocompatibility complex products, including monomers, tetramers, biotinylation, and fluorescent labeling. Our MHC products are mammalian-expressed and bioactivity-verified. Order an off-the-shelf MHC product or request a custom MHC complex.

Customize your MHC
MHC Monomers & Tetramers FAQs
MHC monomers consist of a single MHC-peptide complex and are typically used for coating plates in ELISA, SPR, or biophysical assays. MHC tetramers are formed by conjugating four MHC monomers to streptavidin, which enhances avidity and allows for the detection of antigen-specific T cells via flow cytometry. For functional assays and T cell staining, tetramers are the preferred format due to their multivalent nature.
MHC class I molecules are typically used for presenting intracellular peptides to CD8+ cytotoxic T cells, while class II molecules present extracellular-derived peptides to CD4+ helper T cells. Your choice should depend on the immunological context of your study—e.g., tumor antigen presentation (class I) or vaccine development and helper T cell profiling (class II).
Peptide-ready MHCs (prMHCs) are stabilized MHC molecules supplied without a bound peptide. They are designed to allow easy, in-lab loading of custom antigenic peptides, offering flexibility in screening and experimental design. In contrast, pre-loaded MHCs are supplied with specific peptides already bound and validated.
Yes. Our fluorescent-labeled MHC tetramers (e.g., PE, APC, FITC) are designed for flow cytometry and FACS applications. These products are validated for use in identifying and sorting antigen-specific T cells from human or mouse samples. Ensure compatibility with your fluorophore and cytometer settings.
We offer a broad range of human and mouse MHC class I alleles, including HLA-A02:01, HLA-B07:02, HLA-C*07:02, and mouse H-2Kb, among others. For less common alleles or non-classical MHCs, we recommend using our Custom MHC Services.
Most MHC products are available with C-terminal His-tags, biotinylation, or fluorescent labels for assay compatibility. These tags facilitate detection, immobilization, or tetramer assembly depending on the intended application.
Each product undergoes ELISA, SPR, and/or FACS validation to confirm specific peptide–TCR binding and structural integrity. We assess EC50, KD values, and batch reproducibility using techniques such as SPR and BLI to ensure performance across applications.
Yes. We offer custom MHC services that allow you to request MHC monomers or tetramers loaded with your peptide of interest. Alternatively, you can use a peptide-ready MHC to load your own peptide in-house using standard tetramerization protocols.
Lyophilized MHCs should be stored at –20°C or below. Reconstitution should be done using sterile, peptide-compatible buffers. Detailed reconstitution instructions and storage guidelines are provided in the product datasheet or upon request.
Our MHC products are used in TCR affinity testing, antigen-specific T cell detection, blocking assays, drug screening, and immune profiling. With formats available for ELISA, SPR, flow cytometry, and in vivo models, they support diverse workflows in cancer immunology and infectious disease research.