STEAP1, the Next Blockbuster Target for Prostate Cancer
By Tingxu Chen
Prostate cancer (PCa) is the second most common cancer among men, with a mortality rate of approximately 2.3%. According to the American Cancer Society, around 35,000 men die of prostate cancer each year in the United States, and nearly 400,000 deaths occur globally[1], with most deaths occurring in men over the age of 75, and in advanced stages such as advanced prostate cancer and metastatic prostate cancer. In pathology reports, this is often described as prostate carcinoma or prostate cancer prostatic dis (prostatic disease).
Among all PCa subtypes, metastatic castration-resistant prostate cancer (mCRPC) is the most challenging to treat, with a median survival time of less than three years for patients with metastatic castration resistance. Currently, the prostate-specific membrane antigen (PSMA) is the predominant diagnostic and therapeutic target for mCRPC, used in imaging and treatments such as radionuclide therapy (e.g., [^177Lu]Lu-PSMA Pluvicto). However, issues such as drug resistance, disease recurrence, and low PSMA expression in part of the patient populations also limit its efficacy, which highlights the urgent need for targeted therapies targeting advanced prostate cancer.
The STEAP (Six Transmembrane Epithelial Antigen of the Prostate) protein family is a group of tumour-associated antigens first discovered in 1999. Within this protein family, STEAP1 was identified as a prostate-associated transmembrane epithelial antigen with high STEAP1 expression on prostate cancer cells and very low signal in most normal tissues. Studies over the past two decades have consistently shown that STEAP1 is closely associated with PCa progression—including bone metastasis and castration resistance—and that its expression level positively correlates with patients with poor prognosis, in human prostate tumors, which makes STEAP1 a compelling steap1 prostate cancer target for cancer immunotherapy.
At the 2024 AACR Annual Meeting, Amgen presented the promising Phase 1 clinical trials data for AMG509 (Xaluritamig), a bispecific T-cell engager (TCE) targeting STEAP1. In patients with metastatic castration resistant prostate (mCRPC) cancer, 49% experienced a ≥50% reduction in PSA levels, and 28.4% achieved a ≥90% reduction. The objective response rate (ORR) in the high-dose group was 41%, with a disease control rate (DCR) of 79% by standard response evaluation criteria. This marks the first STEAP1-targeted therapy to demonstrate robust objective tumor responses in resistant prostate cancer mCRPC, reinforcing STEAP1 as a high-value steap1 prostate cancer target.
STEAP1 is a six-transmembrane protein with both its N- and C-termini located in the cytoplasm. Unlike other STEAP family members, STEAP1 lacks an NADPH-binding FNO domain in its N-terminus. It cannot independently catalyze Fe³⁺ or Cu²⁺ reduction 222, requiring interaction with other STEAP proteins[2] and reflecting a potential enzymatic function that depends on complex formation. A truncated isoform, STEAP1B, is considered to be involved in metal ion metabolism and the regulation of malignant tumor phenotypes.
The structure of STEAP1; STEAP1 is highly expressed in prostate cancer tissues [2]
STEAP1 structure and expression
STEAP1 is predominantly expressed at cell–cell junctions, especially in prostate secretory epithelial cells, maintaining cellular redox balance and regulating proinflammatory cytokine signaling. In addition, STEAP1 also functions as one of several cell surface antigens that mark prostate tumor cells in tissue. In cancers such as bladder and prostate cancer, and other solid tumors, STEAP1 is highly expressed and critical for tumor proliferation and metastasis. In mCRPC, the STEAP1 expression is driven by androgen receptor (AR) signaling. Even under low-testosterone, castration-resistant conditions, after extended androgen deprivation therapy—AR splice variants (e.g., AR-V7) can continue to drive STEAP1, supporting tumor progression and survival of tumor cells.
Overexpression of STEAP1 suppresses CD8⁺ T cells infiltration, enhances immunosuppressive microenvironment, and accelerates disease progression. This aligns with emerging data on the tumor immune microenvironment, where tumor-intrinsic drivers like STEAP1 interact with immune checkpoint inhibitors, T-cell signalling, and antigen presentation via major histocompatibility complex (MHC) to limit t cell activation against cancer cells. Preclinical work has also indicated that STEAP1 may contribute to radiotherapy and chemotherapy resistance, likely via reduced ferroptosis and adaptation of vivo tumor growth patterns.
Mechanism of action of STEAP1 [3]
Advances in STEAP1-Targeted Therapies
Currently, the drug development targeting STEAP1 is under rapid progressing, with antibody-drug conjugates (ADCs) and T-cell engagers (TCEs) emerging as the leading targeted therapies. Both single- and dual-antigen targeting strategies are under investigation, primarily for advanced prostate cancer and mCRPC, with additional work in other human cancer indications.
ADCs:
Adcentrx’s ADRX-0405 employs a high drug-to-antibody ratio (DAR = 8), protease-cleavable linkers, and stable conjugation chemistry to enhance payload delivery and ADC stability in STEAP1-positive prostate tumor cells and other tumor antigens–bearing tumor cells.
DAC Biotechnolog’s DXC008 and AbbVie’s ABBV-969 are dual-targeting ADCs targeting both STEAP1 and PSMA (prostate-specific membrane antigen), designed to manage biochemical recurrence and resistance in mCRPC and toimprove overall antitumor efficacy. Both candidates are currently in Phase 1 trials.
T-cell–engaging therapies (TCEs):
Amgen’s AMG509 (Xaluritamig) is a leading STEAP1-targeted TCE, utilizing a 2+1 bispecific format to bind two STEAP1 molecules and one CD3 molecule. On T cells, creating focused T cell activation within the tumor microenvironment. This format has shown potent anti-tumor efficacy in cancer res–reported datasets for patients with metastatic castration resistance and liver metastases. Preclinical CAR T cell therapy programs are also emerging, including steap1 CAR T cells that introduce a steap1 chimeric antigen receptor into an antigen receptor T cell (T lymphocyte). These CAR T cell constructs are being evaluated for antitumor efficacy in STEAP1-positive models and represent a next wave of cell therapy beyond PSMA-directed CAR T cell therapy. The clinical trial has proceeded directly from Phase 1 to Phase 3, reflecting its strong clinical and commercialization potential [4].
BC261, developed by Memorial Sloan Kettering Cancer Center, uses a 2+2 IgG-[L]-scFv structure targeting a conserved extracellular domain of STEAP1. This format may offer more consistent binding and efficacy across species. BC261 is currently in preclinical development [5].
Diagnostic Imaging and Surgical Applications
Beyond therapeutics, STEAP1 also holds promise for clinical imaging and precision surgery. In a recent study, scientists developed a fluorescent imaging probe using extracellular vesicles engineered to target the STEAP1 protein. This probe significantly improved the imaging accuracy and contrast, reducing the positive surgical margin rate from 100% to 0% in murine models. It also extended postoperative survival, offering a valuable reference for precision-guided prostate cancer surgery [7].
List of therapeutic drugs targeting STEAP1 [2]
Structure of Amgen's TCE drug AMG-509 [4]
During AMG-509 treatment, the patient's serum PSA level decreased significantly [4]
KACTUS STEAP1 Reagents for Translational Work
Due to its tumor-specific high expression, significant role in therapy resistance, and favorable drugability, STEAP1 has become one of the most promising therapeutic targets in prostate cancer. Drugs targeting STEAP1 could fill critical gaps in the treatment landscape, particularly for patients resistant to AR inhibitors, and may emerge as the next major target after PSMA.
By leveraging the recombinant multi-transmembrane protein technology platform, KACTUS has developed a full-length, conformationally correct STEAP1 protein in VLP format with stable and robust biological activity. These reagents are designed for discovery and validation against prostate cancer cells and LNCaP prostate cancer cells, as well as models of prostatic intraepithelial neoplasia lesions and prostate tumor cells, helping teams link early assay readouts to potential survival benefit in future clinical trials.
Product Data:
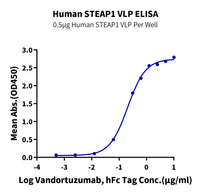
Immobilized Human STEAP1 VLP at 5 μg/ml (100 μl/well) on the plate. Dose response curve for Vandortuzumab with the EC 50 of 0.21 μg/ml determined by ELISA.
Product List:
|
Part Number |
Information |
|
Human STEAP1 VLP |
|
|
Biotinylated Human STEAP1 VLP |
|
|
Cynomolgus STEAP1 VLP |
FAQs About STEAP1 as a Prostate Cancer Target
1. How does STEAP1 compare with PSMA and other prostate antigens?
PSMA (prostate-specific membrane antigen) remains the primary antigen of the prostate used in imaging and targeted therapy for mCRPC, but limited expression in some prostate cancer populations and emerging resistance have created a need for alternatives. STEAP1 offers:
-
High steap1 expression in advanced prostate cancer, often maintained in castration resistant prostate cancer
-
Minimal expression in normal tissues, improving the therapeutic window
-
Presence on cell surface antigens of prostate cancer cells and human prostate tumors, including some prostatic intraepithelial neoplasia lesions
Alongside STEAP1 and PSMA, prostate stem cell antigen and prostatic acid phosphatase are also under investigation, but current clinical trials data position STEAP1 as one of the more attractive next-generation tumor antigens for targeted therapies.
2. What role does STEAP1 play in disease progression and the tumor microenvironment?
In preclinical and translational studies, STEAP1 has been linked to:
-
Increased proliferation and metastatic behaviour of tumor cells in prostate carcinoma.
-
Reduced CD8⁺ t cells infiltration and an immunosuppressive tumor microenvironment.
-
Changes in cytokine signalling in the tumor immune microenvironment, affecting cell activation and response to immune checkpoint inhibitors.
These features make STEAP1 a marker of both tumor progression and immune escape, which is why it is now widely discussed in the context of cancer immunotherapy for metastatic castration resistant prostate disease.
3. Which STEAP1-targeted therapeutic strategies are under investigation?
The main modalities being investigated include:
-
Antibody drug conjugates directed at STEAP1 alone or together with PSMA, evaluated for dose exploration, safety, and objective tumor responses in mCRPC
-
Bispecific T cell engagers that connect STEAP1 on cancer cells with CD3 on T cells, driving focused t cell activation
-
Experimental CAR T cell therapy platforms, where STEAP1 CAR t cells bearing a STEAP1 chimeric antigen receptor (STEAP1-specific chimeric antigen receptor) are tested for antitumor efficacy in preclinical models
These approaches are being benchmarked not only in prostate indications but also in other solid tumors, with some comparison to established T-cell therapies in large B-cell lymphoma and other settings.
4. Is STEAP1 relevant beyond the prostate?
Yes. While initially defined as a prostate-associated antigen, STEAP1 has been reported in several human cancer types:
-
Subsets of colorectal cancer
-
Selected lung adenocarcinoma cohorts
-
Metastatic lesions in soft tissue and liver metastases
In these contexts, STEAP1 behaves as one of several tumor antigens on cancer cells, supporting its broader potential beyond the prostate while still requiring careful selection of imaging patients and therapy candidates.
5. How do KACTUS STEAP1 tools support rigorous research and translation?
KACTUS focuses on providing reagents suitable for both exploratory work and later-stage validation:
-
Full-length STEAP1 in VLP format designed to preserve native conformation on cell surface antigens.
-
Use in functional assays with LNCaP prostate cancer cells, primary prostate tumor cells, and engineered lines modelling advanced metastatic prostate cancer.
-
Application across discovery, candidate selection, and translational studies that feed into clinical trials of targeted therapies, cell therapy, and cancer vaccines.
By combining high-quality protein tools with data packages and publication-mapped references, the platform supports methodologically sound projects that can be evaluated against standard clinical endpoints and response evaluation criteria.