71 products
Background
Chimeric Antigen Receptor T-Cell Immunotherapy (CAR-T) is a novel immunotherapy that has achieved success in the treatment of various hematological malignancies.
The concept behind CAR-T therapy is to empower the immune system to attack cancer cells. In short, a patient's T cells are isolated and genetically engineered to express a chimeric antigen receptor (CAR), which recognizes specific tumor-associated antigens, such as CD19, a cell surface protein highly expressed on malignant B cells. Upon recognizing the antigen on tumor cells, CAR-T cells initiate a series of immune responses to kill the tumor cells. With an increasing number of CAR-T drugs being approved for marketing (such as Kymriah and Yescarta), CAR-T cell therapy has entered a period of rapid development, demonstrating immense potential.
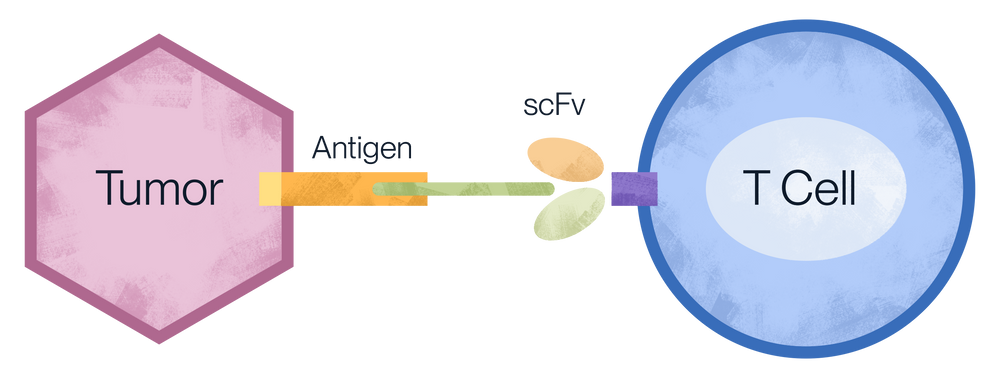
CAR-T cell binding tumors mechanism.
Comprehensive Catalog of CAR-T Target Proteins
Leveraging our unique SAMS™ protein engineering and expression platform, KACTUS has developed a series of high-quality CAR-T target protein products, covering a wide range of popular targets such as CD19, BCMA, and MSLN. We also offer various modifications including site-specific labeling, fluorescent labeling, and biotinylation, flexibly meeting diverse research and development needs such as CAR-T affinity assays. If you don't see what you're looking for in our catalog, you can always request a custom protein.
Product Features
→ HEK293 Expression
→ His-Avi, hFc, mFc, His, hFc-Avi, etc. protein tags
→ Bioactivity verified (ELISA & SPR)
→ High Purity
→ Human, Mouse, Cynomolgus, Rhesus Macaque, Rat
→ Biotinylated Proteins
Product Applications
→ CAR-T immunity studies
→ Antibody screening (such as scFv, single domain antibody, etc.)
→ CAR-T affinity studies
Featured CAR-T Targets
Product Validation Data
KACTUS offers a wide range of fluorescent-labeled target proteins that streamline CAR detection by eliminating the need for secondary antibodies, reducing background staining, and increasing efficiency. Our portfolio includes popular target antigens relevant to current CAR-T research, all with high bioactivity, uniformity, and batch-to-batch consistency.
Leveraging our advanced protein expression platform, KACTUS also provides a comprehensive solution for protein customization. We offer various modifications such as site-specific labeling, fluorescent labeling (FITC, PE, APC, and others), and biotinylation, to meet various research requirements such as CAR-T affinity testing.
FITC-Compatible Human CD19
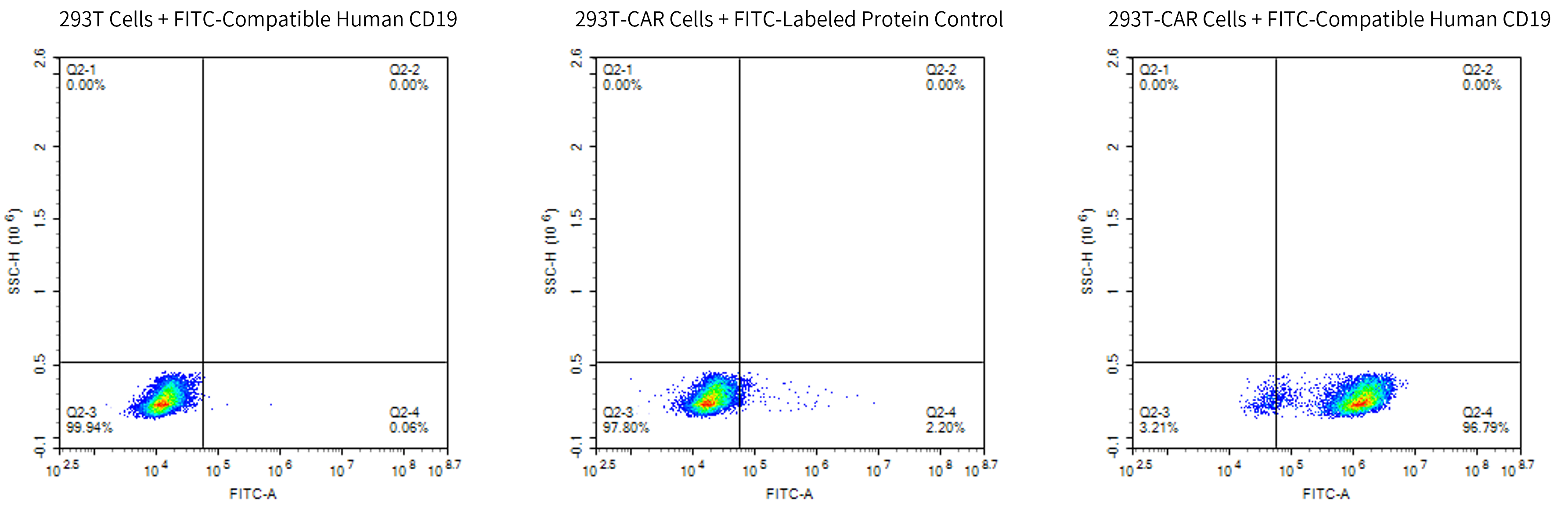
FACS data demonstrating that 100 µl of FITC-Compatible Human CD19 (His Tag) (10 µg/ml) can specifically bind to 1×106 Anti-CD19 CAR T cells, resulting in a positive detection rate greater than 96%. FITC-labeled irrelevant protein (100 µl, 10 µg/ml) was used as a negative control.
PE-Labeled Human EGFRVIII
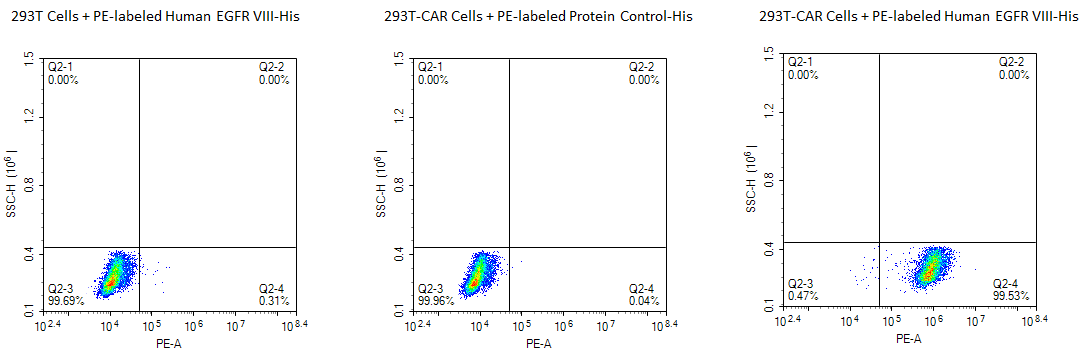
FACS analysis of Anti-EGFRVIII CAR Expression. 293T cells were transfected with anti-EGFRVIII-scFv and His tag. Cells were incubated with 5 µg/ml PE-Labeled Human EGFRVIII (His-Avi Tag) and PE-labeled protein control. Non-transfected 293T cells and PE-labeled protein control were used as negative controls. The PE-labeled Human EGFR VIII specifically binds to Anti-EGFR VIII-CAR T cells, with a positive detection rate of over 99%.
Biotinylated Human MSLN/Mesothelin
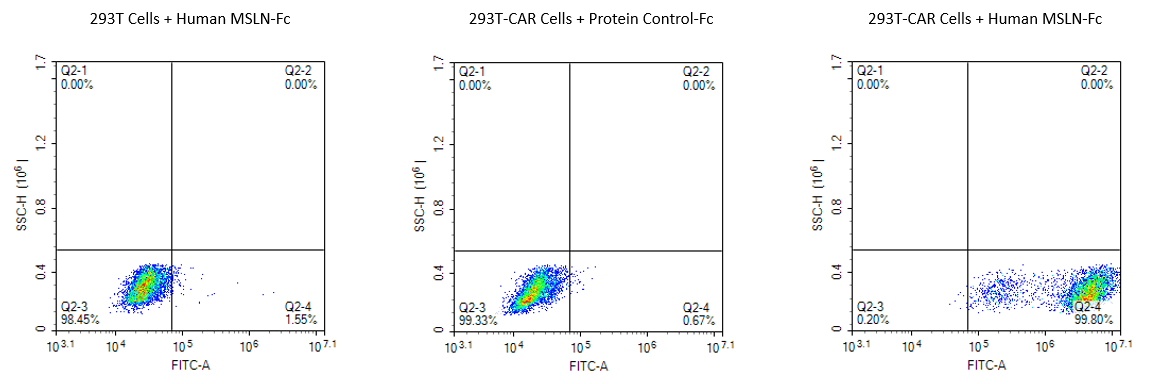
FACS analysis of Anti-MSLN-CAR expression. 293T cells were transfected with anti-MSLN-scFv and Fc tag. These cells were stained with 5 µg/ml FITC-Labeled Human MSLN (296-580), Fc Tag, and a FITC-labeled protein control. Non-transfected 293T cells and FITC-labeled protein control were used as negative controls. FITC-labeled SA was used to confirm that Biotinylated Human MSLN (hFc-Avi Tag) can specifically bind to Anti-MSLN-CAR 293T cells, resulting in a positive rate greater than 99%.

Customize your protein

About KACTUS

SAMS™ Technology Platform

Quality Control
Related Products & Information
CAR-T Products & Services
Gene Editing Enzymes





