MSLN Targeted Drugs Entering the Fast Lane
By Yujiao Zhang
Since December 2023, there have been several positive updates about drugs targeting MSLN. On December 19th, RemeGen announced that the U.S. FDA has approved the Phase II clinical trial application of RC88 for treating platinum-resistant recurrent epithelial ovarian cancer, fallopian tube cancer, and primary peritoneal cancer patients. This drug will be tested in multiple countries and regions including the United States, China, and the European Union. On December 15th, Nona Biosciences (a subsidiary of Harbour BioMed) signed a global clinical development and commercialization authorization agreement with Seagen (a subsidiary of Pfizer) for HBM9033. As part of the agreement, Nona Biosciences will receive a $53 million upfront payment and near-term payments, as well as up to $1.05 billion in milestone payments and royalty fees.
Expression and Function of MSLN
MSLN (Mesothelin) is a protein that is anchored to the membrane by GPI, without any transmembrane or intracellular regions. This protein can be cleaved by enzymes like Furin into two segments: a soluble form of approximately 31 kDa known as Megakaryocyte Potentiating Factor (MPF), and a mature form of around 40 kDa which remains tethered to the membrane. The mature form of MSLN can also be shed to produce soluble MSLN and truncated MSLN that remains attached to the membrane. 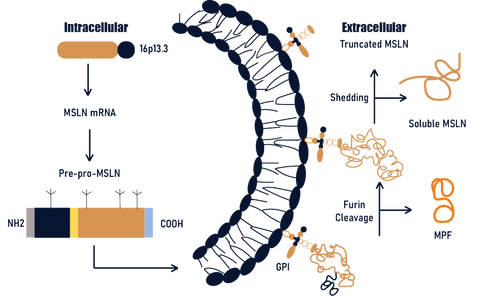
Figure 1: Structure of MSLN [1]
MSLN is a tumor-associated antigen (TSA). Unlike typical TSAs, MSLN is expressed only in limited tissues, such as the peritoneum, pleura, and mesothelial cells, leading to lower nonspecific toxicity, making it an ideal drug target. MSLN is highly expressed in common cancers such as malignant pleural mesothelioma, pancreatic cancer, ovarian cancer, colorectal cancer, and other tumor tissues. However, its expression level varies across different tumors. Additionally, MSLN can participate in the growth, adhesion, and migration of different tumor cells through various signaling pathways. Therefore, in clinical practice, it is necessary to flexibly consider the selection of indications and the types of combination therapies.
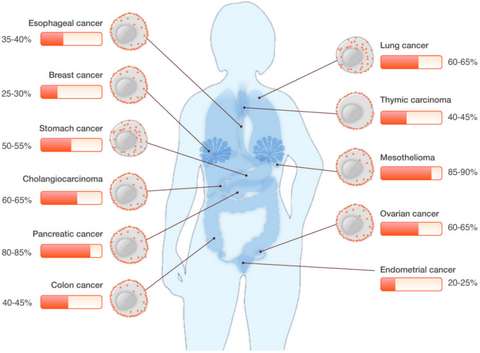
Figure 2: Expression of MSLN in different cancers [2]
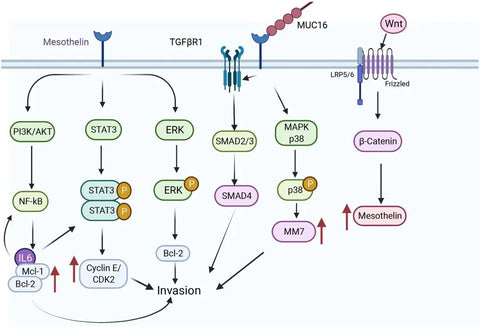
Figure 3: MSLN-related signaling pathways [3]
Role of Mesothelin in Cancer
MSLN plays a functional role beyond its role as a surface biomarker. It contributes to tumour cell adhesion, invasion, and interaction with the tumour microenvironment. One of the key mechanisms involves binding to MUC16 (CA125), thereby facilitating cell-cell adhesion and supporting metastatic spread, particularly in ovarian and pancreatic malignancies.
In many tumor tissues, elevated MSLN expression is associated with increased tumour aggressiveness. Its localisation on the cell surface also makes it highly accessible to targeted biologics, including monoclonal antibody therapies and antibody-drug conjugates, which rely on stable antigen exposure for effective binding.
Molecular Mechanisms of MSLN in Tumor Progression
MSLN contributes to tumour progression through multiple signalling pathways that regulate proliferation, survival, and metastasis. It has been linked to activation of pathways such as MAPK and PI3K/AKT, which drive tumour growth and resistance to apoptosis.
In addition, MSLN-mediated interactions can promote epithelial–mesenchymal transition (EMT), enhancing the invasive capacity of cancer cells. These effects are particularly relevant in advanced solid tumors, where disease progression is driven by both intrinsic signalling and microenvironmental factors.
Sustained expression of MSLN in tumor tissues also supports tumour cell survival under stress conditions, reinforcing its value as a therapeutic target in precision oncology strategies.
Progress of Targeted Drugs for MSLN
There are various types of drugs targeting MSLN, including monoclonal antibodies, bispecific antibodies, ADCs, CAR-T/NK therapies, and cancer vaccines, etc. Among them, Bayer's ADC drug BAY94-9343 (Anetumab ravtansine), which is composed of a fully human monoclonal antibody MF-T and microtubule inhibitor DM4 conjugate, has a drug-antibody ratio (DAR) of 3.2, and exhibits bystander killing effect. It has shown a good clinical disease control rate (DCR) and safety profile, making it the fastest progressing ADC currently. However, Eisai's monoclonal antibody MORAb-009 (Amatuximab) and Genentech's ADC DMOT4039A both failed in phase II trials. Despite Harpoon's trispecific antibody HPN536 attempting to salvage the situation, MSLN antibody drugs still face skepticism, gradually leaning towards cell therapies.
The disclosed CAR-T therapy BZD1901 from Shanghai Cell Therapy Group can secrete PD-1 nanobodies, achieving a 100% DCR in mesothelioma (including 1 complete response), with a duration of response (DOR) of 24 months and an objective response rate (ORR) of 63.6%. No serious adverse events (SAE) were reported, indicating the huge potential of CAR cell therapy for targeting MSLN. Additionally, MCY-M11 developed by MaxCyte utilizes mRNA non-viral technology, designs CAR based on macrophages, and recruits T cells, offering better penetration and targeting efficiency in advanced solid tumors. Furthermore, the TCR-T drug TC-210 (Gavo-cel), developed by TCR2, showed an initial clinical ORR of 38% and a DCR of 100%.
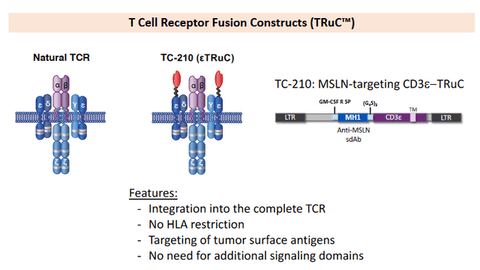
Figure 4: Mechanism of action of TC-210 [4]
Challenges in MSLN-targeted Therapy
Despite strong interest in MSLN-targeted approaches, several challenges remain in clinical development. One major limitation is antigen heterogeneity, where varying expression levels across tumor tissues can reduce treatment consistency.
Shedding of MSLN into circulation presents another barrier, as soluble antigen can interfere with antibody binding and reduce effective targeting at the cell surface. This directly affects therapies such as antibody-drug conjugates, which rely on stable antigen engagement for payload delivery.
There is also a need to minimise off-target effects in normal tissues, despite MSLN expression being relatively restricted. Addressing these challenges requires improved epitope selection, better control of antigen-binding affinity, and optimisation of drug design.
Design of Membrane-Proximal Epitope for MSLN
MSLN exists in different forms, and when designing targeting sites, considerations need to be given to hindrances from soluble MSLN and competition from MUC16 binding. Additionally, continuous shedding of MSLN by tumor cells leads to further release of antibodies binding to most regions of MSLN. Therefore, many drugs set their epitopes at the membrane-proximal end with crystal structure [5][6], such as Nona Biosciences's ADC HBM9033. This approach not only enhances the cell's ability to establish contact but also improves the therapeutic window while reducing adverse reactions.
Figure 5: MSLN-targeted drugs designed with Membrane-Proximal Epitopes [7][8]
List of MSLN-targeted drugs entering clinical trials:
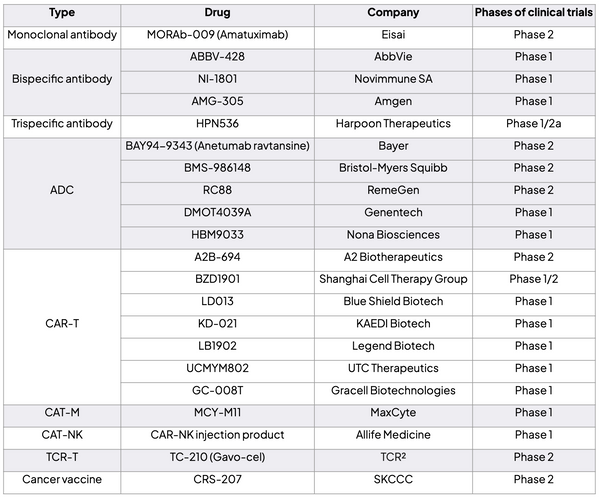
Table 1: Selected MSLN-targeted drugs in the clinic
Prognostic Value of MSLN in Cancer
MSLN expression has been investigated as a prognostic biomarker across multiple cancer types. Elevated levels are often associated with poor clinical outcomes, including reduced overall survival and increased tumour progression rates.
In cancers such as mesothelioma and ovarian cancer, high MSLN expression correlates with tumour burden and disease stage. This makes it a useful indicator for patient stratification and treatment planning.
From a therapeutic perspective, measuring antigen concentration and distribution across tumor tissues can help guide the selection of targeted treatments, including drug conjugates and other biologics designed for precision targeting.
Recombinant MSLN protein
MSLN is an attractive target for many cancer therapies due to its limited physiological expression and overexpression characteristics in various tumors. KACTUS has developed high-quality recombinant MSLN proteins from different species and with different tags. In particular, we can provide various membrane-proximal domain proteins. All our products undergo rigorous quality testing and can be used in various stages of research and development such as immunization, antibody screening, mechanism validation, etc. This helps support the development of MSLN-targeted drugs.
Product Validation Examples

|
 |
| Figure 6: Human MSLN (Catalog number MSL-HM181) can bind to Biotinylated Human CA125 with an EC50 of 11.7 ng/ml, as determined by ELISA. | Figure 7: Cynomolgus MSLN (Catalog number MSL-CM180) can bind to Anti-MSLN Antibody with an EC50 of 14.1 ng/ml, as determined by ELISA. |
FAQ's
1. Why is MSLN considered a strong target in cancer therapy?
MSLN is a tumor-associated antigen with limited expression in normal tissues, but high expression in solid tumors, making it suitable for targeted therapies.
2. How do antibody drug conjugates work in MSLN targeting?
Antibody drug conjugates combine a targeting antibody with a cytotoxic payload, enabling selective delivery to tumor tissues while reducing systemic toxicity.
3. What role does antigen concentration play in therapy effectiveness?
Higher antigen concentration on the cell surface improves binding efficiency and increases the likelihood of therapeutic success, especially for targeted biologics.
4. Why are membrane-proximal epitopes preferred?
They improve binding stability, reduce interference from soluble proteins, and support better drug internalisation in cancer cells.
5. Why are mouse models used in MSLN research?
Mouse models allow preclinical evaluation of drug safety, efficacy, and targeting behaviour before clinical trials in humans.
Available MSLN Proteins
|
Catalog Number |
Product Information |
|
Human MSLN (M593V), C-His |
|
|
Human MSLN (296-598), C-His |
|
|
Human MSLN (296-591), C-His |
|
|
Human MSLN (487-598), C-His-Avi |
|
|
Human MSLN (487-598), C-hFc |
|
|
Cynomolgus MSLN (406-690), C-His |
|
|
Biotinylated Cynomolgus MSLN (406-690) (Primary Amine Labeling), C-His |
|
|
Cynomolgus MSLN (296-598), C-His |
|
|
Cynomolgus MSLN, C-hFc |
|
|
Mouse MSLN, C-His |
|
|
Biotinylated Mouse MSLN (Primary Amine Labeling), C-His |
Click the catalog number for product details.
References
[1] https://2023.igem.wiki/patras-med/description
[2] Morello A, Sadelain M, Adusumilli PS. Mesothelin-Targeted CARs: Driving T Cells to Solid Tumors. Cancer Discov. 2016 Feb;6(2):133-46.
[3] Li D, Lin S, Hong J, Ho M. Immunotherapy for hepatobiliary cancers: Emerging targets and translational advances. Adv Cancer Res. 2022;156:415-449.
[4] https://www.tcr2.com/
[5] Hatterer E, Chauchet X, Richard F, Barba L, Moine V, Chatel L, Broyer L, Pontini G, Bautzova T, Juan F, Calloud S, Bosson N, Charreton M, Masternak K, Buatois V, Shang L. Targeting a membrane-proximal epitope on mesothelin increases the tumoricidal activity of a bispecific antibody blocking CD47 on mesothelin-positive tumors. MAbs. 2020 Jan-Dec;12(1):1739408.
[6] Novel mesothelin antibodies enable crystallography of the intact mesothelin ectodomain and engineering of potent, T cell-engaging bispecific therapeutics.
[7] Grasso L, Jiang Q, Hassan R, Nicolaides NC, Kline JB. NAV-003, a bispecific antibody targeting a unique mesothelin epitope and CD3ε with improved cytotoxicity against humoral immunosuppressed tumors. Eur J Immunol. 2023 Aug;53(8):e2250309.
[8] Liu X, Onda M, Watson N, Hassan R, Ho M, Bera TK, Wei J, Chakraborty A, Beers R, Zhou Q, Shajahan A, Azadi P, Zhan J, Xia D, Pastan I. Highly active CAR T cells that bind to a juxtamembrane region of mesothelin and are not blocked by shed mesothelin. Proc Natl Acad Sci U S A. 2022 May 10;119(19):e2202439119.















