The culprit of cancer metastasis - uPAR-uPA
By Mallory Griffin
Tumor invasion and metastasis are key risk factors for tumor recurrence and patient death. Tumor invasion occurs when changes in cell-cell adhesion and cell-matrix adhesion allow malignant cells to invade surrounding tissue. uPAR/uPA is involved in the degradation of tumor extracellular matrix (ECM), tumor angiogenesis, and tissue infiltration. Thus, uPAR-uPA has attracted attention in recent years as a target for therapeutic drugs.

uPAR is a high-affinity receptor for urokinase-type plasminogen activator (uPA, also known as PLAU), which is anchored to the cell membrane by glycosylphosphatidylinositol (GPI). The extracellular domain of uPAR includes the D1, D2, and D3 domains. The D1-D2 junction can be cleaved to generate D1 and D2-D3, where D1 alone can still bind to uPA, but with at least a 1500-fold decrease in affinity compared to intact uPAR. D2-D3 has the same chemotactic properties as uPA and can combine with GPCR protein FPRL1 to transmit chemotactic signals. uPAR can also be detached from the membrane as a whole to form soluble uPAR (suPAR), which is considered to be an indicator of tumor prognosis. As a ligand, uPA is initially an inactive zymogen, pro-uPA, which can be activated after binding to uPAR to form a double-chain structure linked by active disulfide bonds.

Figure 1: Schematic diagram of uPAR structure [1]
The binding of uPAR and uPA leads to the activation of plasminogen and the degradation of various ECM proteins. When the ECM junctions are no longer tight, tumor cells can begin to migrate and release ECM-related growth factors. These enhance the expression of uPAR and uPA, inducing tumor angiogenesis and epithelial-mesenchymal transition. In addition, uPAR/uPA can regulate cell proliferation and prevent apoptosis, which plays an important role in the occurrence and development of tumors. In addition, uPAR can combine with integrin α5β1, α3β1, or αvβ3 with the assistance of vimentin or fibronectin to form a complex, thereby activating the downstream pathways of integrins such as Ras–MAPK, PI3K–Akt and JAK–STAT, enhancing tumor cell proliferation and migration.
Numerous studies have confirmed that uPAR-uPA is closely related to a variety of malignant tumors. Therapies can target various steps in the mechanism of action (Figure 2). Currently, there are no drugs targeting uPAR-uPA on the market, but there are many being researched. For example, a novel antibody targeting uPAR developed by Lanzhou University can be combined with PD-1 antibody to treat diffuse gastric cancer (DGC), significantly prolonging the survival of model mice. The CAR-T therapy developed based on this antibody effectively kills DGC organoids [2]. Presently, they have applied for domestic and foreign patents, with good clinical transformation prospects. Other potential uPAR inhibitors include uPAR (Ser88-Tyr92) peptide and MNPR 101 (ATN-658) developed by Monopar Therapeutics. In addition, uPAR is used in aging-related research [3].
uPA inhibitors currently include the drug Upamostat, developed by the German company Wilex, which was introduced by Lingsheng Medical. The hydrogen sulfate capsule, which has been combined with gemcitabine hydrochloride for patients with locally advanced/metastatic pancreatic cancer, is currently in clinical phase I/II. Upamostat is expected to become the world's first anti-pancreatic cancer drug targeting uPA.
It is also worth mentioning that uPAR and uPA can function independently of each other. Particularly uPAR, which is a co-receptor, is associated with a variety of signaling pathways. This along with the species specificity and structural complexity of uPAR brings about certain challenges in the development of related drugs.

Figure 2. Strategies for targeting uPAR-uPA [4]
KACTUS provides highly active uPAR and uPA proteins.
Through its self-developed innovative recombinant protein production platform, Structure Aided Design and Multiplex Screening (SAMS), KACTUS has launched a series of highly active uPAR-uPA and related proteins. The product line covers different forms of proteins, different species, and various labelings such as biotinylation. The products can be used in various scenarios such as immunization, antibody-drug screening, and antibody affinity research. This will help the development of targeted drugs for uPAR/uPA.
Product Data

Immobilized Human PLAU, His Tag at 0.5μg/ml (100μl/Well) on the plate. Dose-response curve for Human uPAR, hFc Tag with an EC50 of 10.3ng/ml determined by ELISA.

Immobilized Human ITGA5&ITGB1, His Tag at 1μg/ml (100μl/well) on the plate. Dose-response curve for Anti-ITGA5&ITGB1 Antibody, hFc Tag with an EC50 of 17.2ng/ml determined by ELISA.

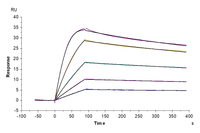
Cynomolgus uPAR, His Tag immobilized on CM5 Chip can bind Cynomolgus PLAU, His Tag with an affinity constant of 0.033nM as determined in SPR assay (Biacore T200).
KACTUS uPAR/uPA Series Products
|
Catalog Number |
Description |
|
Human PLAU (pro form), His Tag |
|
|
Human PLAU (active form), His Tag |
|
|
Biotinylated Human PLAU (active form), His-Avi Tag |
|
|
Cynomolgus PLAU, His Tag |
|
|
PAR-HM401 |
Human uPAR, His-Avi Tag |
|
Biotinylated Human uPAR, His-Avi Tag |
|
|
Human uPAR, hFc Tag |
|
|
Cynomolgus uPAR, His Tag |
|
|
Mouse uPAR isoform 1, His-Avi Tag |
|
|
Biotinylated Mouse uPAR isoform 1, His-Avi Tag |
|
|
Human ITGA5&ITGB1, His-Avi Tag |
|
|
Biotinylated Human ITGA5&ITGB1, His-Avi Tag |
|
|
Human ITGAV&ITGB3, His-Avi Tag |
|
|
Biotinylated Human ITGAV&ITGB3, His-Avi Tag |
|
|
Human ITGAV&ITGB3, hFc Tag |
|
|
Mouse ITGAV&ITGB3, His Tag |
|
|
ITG-MM2V3 |
Mouse ITGAV&ITGB3, hFc Tag |
References
[1] Blasi F, Carmeliet P. uPAR: a versatile signaling orchestrator. Nat Rev Mol Cell Biol. 2002 Dec;3(12):932-43. doi: 10.1038/nrm977. PMID: 12461559.
[2] Qin L, Wang L, Zhang J, Zhou H, Yang Z, Wang Y, Cai W, Wen F, Jiang X, Zhang T, Ye H, Long B, Qin J, Shi W, Guan X, Yu Z, Yang J, Wang Q, Jiao Z. Therapeutic strategies targeting uPAR potentiate anti-PD-1 efficacy in diffuse-type gastric cancer. Sci Adv. 2022 May 27;8(21):eabn3774. doi: 10.1126/sciadv.abn3774. Epub 2022 May 25. PMID: 35613265; PMCID: PMC9132454.
[3] Amor C, Feucht J, Leibold J, Ho YJ, Zhu C, Alonso-Curbelo D, Mansilla-Soto J, Boyer JA, Li X, Giavridis T, Kulick A, Houlihan S, Peerschke E, Friedman SL, Ponomarev V, Piersigilli A, Sadelain M, Lowe SW. Senolytic CAR T cells reverse senescence-associated pathologies. Nature. 2020 Jul;583(7814):127-132. doi: 10.1038/s41586-020-2403-9. Epub 2020 Jun 17. PMID: 32555459; PMCID: PMC7583560.
[4] Smith HW, Marshall CJ. Regulation of cell signaling by uPAR. Nat Rev Mol Cell Biol. 2010 Jan;11(1):23-36. doi: 10.1038/nrm2821. PMID: 20027185.